It's a marker you can measure.
And data you can stand behind.
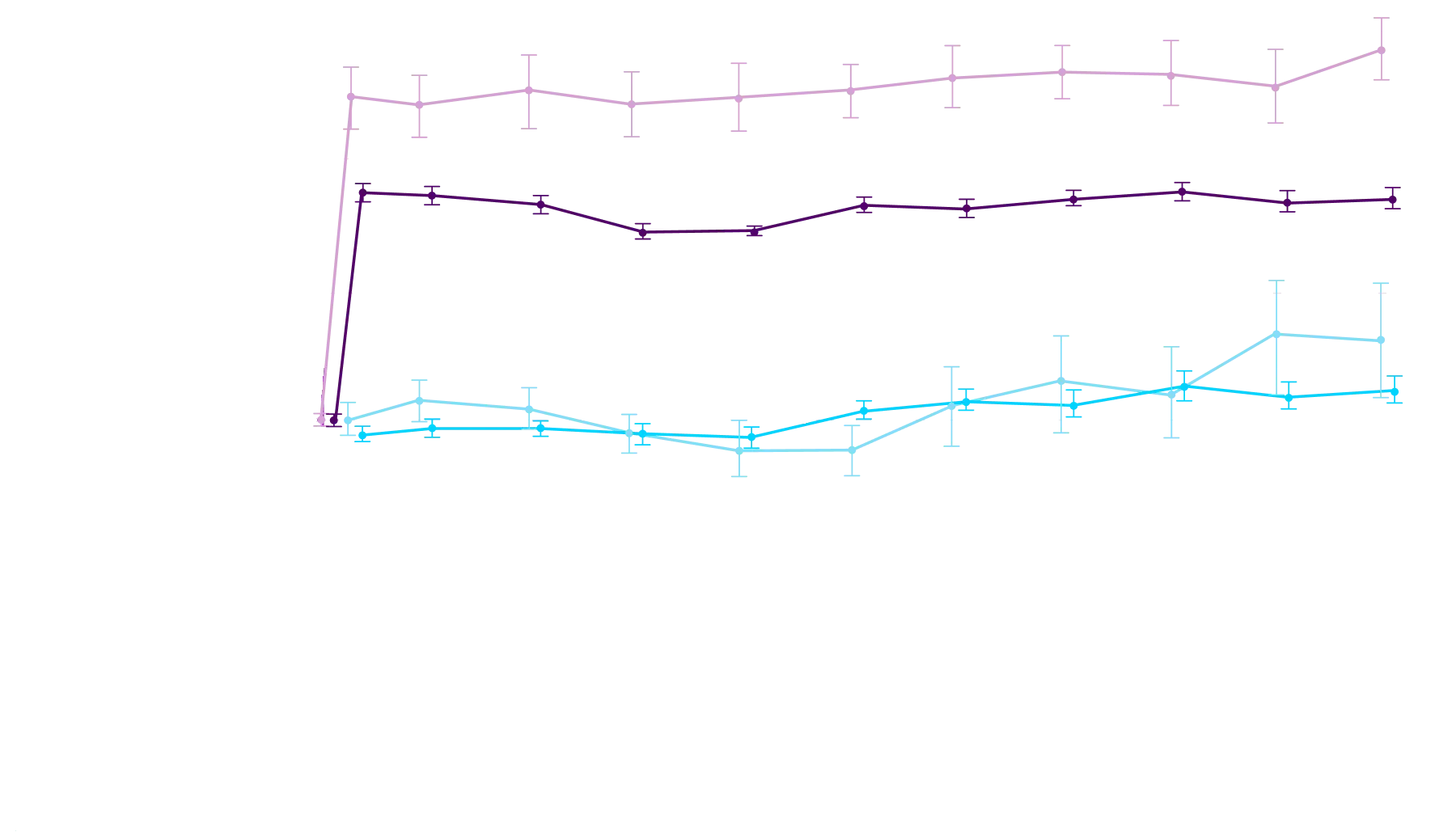
Early increase in serum TTR within
28 days of starting Attruby2
Change From Baseline in Serum TTR Level
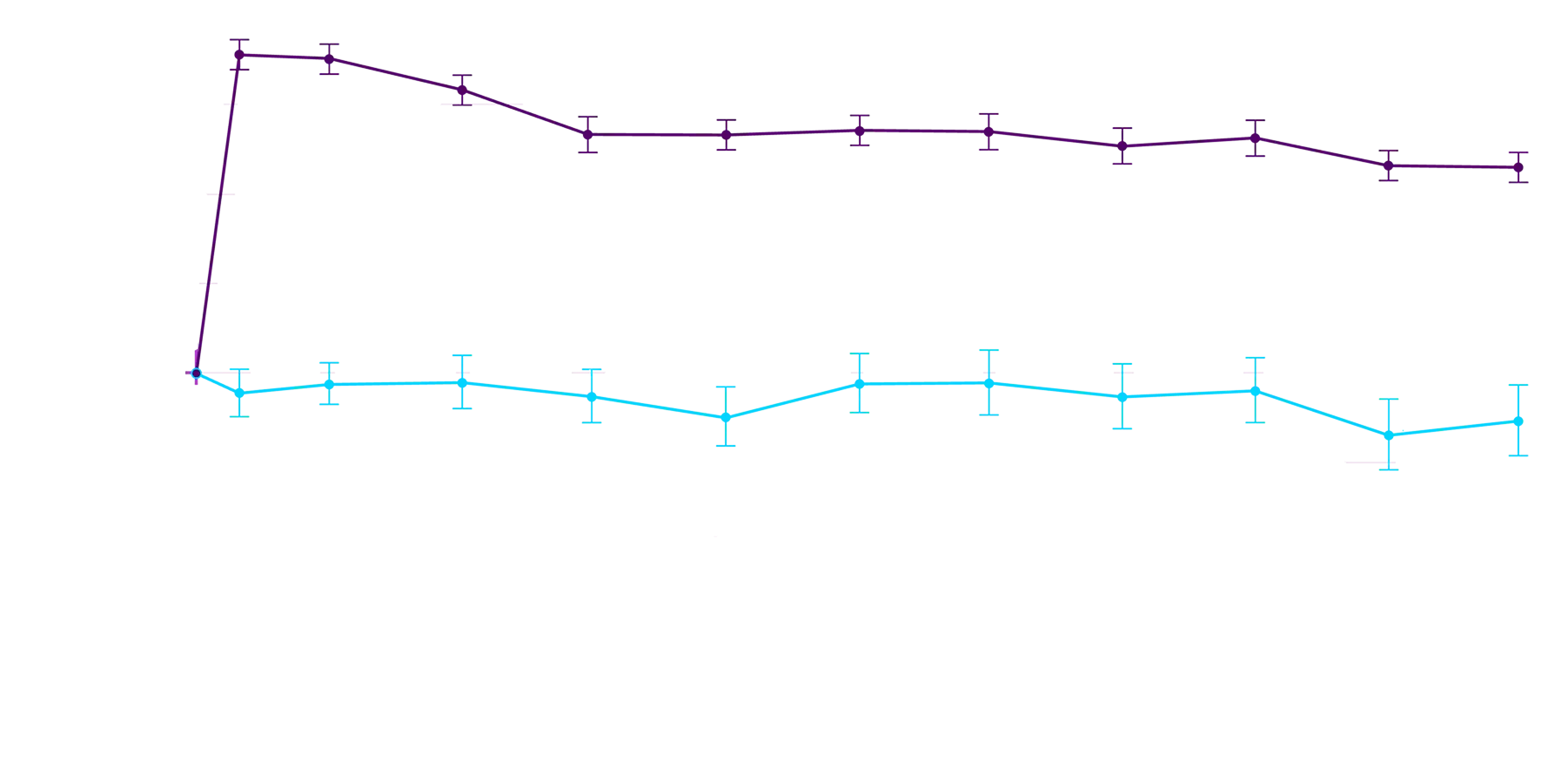

of TTR stability4
Early increases in serum TTR were associated with improved outcomes


 Based on logistic
Based on logistic model prediction5†
 Based on Cox proportional hazards model6‡
Based on Cox proportional hazards model6‡
 Based on Cox proportional
Based on Cox proportional hazards model6‡
These exploratory analyses are based on participants from the ATTRibute-CM phase 3 clinical trial for whom serum TTR levels were available at both baseline and Day 28 and may not fully reflect the broader patient population. Additionally, this effect may not be generalizable to other TTR stabilizers.
†Independent study in 557 patients with ATTR-CM from the ATTRibute-CM study. Univariate analyses in both the overall population and the Attruby-treated population; change in serum TTR remained an independent predictor of ACM even after adjusting for baseline demographic variables, diuretic use, NYHA class, baseline serum TTR, TTR variant or wild-type status, and National Amyloidosis Centre stage.5
‡Analysis of the mITT population of ATTRibute-CM. Combined outcomes of serum TTR CFB to Day 28 and Month 30, and first CVH or cardiovascular mortality, excluding those events that occurred prior to Day 28. Patient stratification was based on treatment group, baseline TTR (<20 or ≥20 mg/dL), and original randomization factors of TTR genotype (wild-type vs variant), NT-proBNP (≤3000 or >3000 pg/mL), and eGFR (<45 or ≥45 mL/min/1.73 m2).6
ACM=all-cause mortality; CFB=change from baseline; CVH=cardiovascular-related hospitalization; CVM=cardiovascular-related mortality; eGFR=estimated glomerular filtration rate; LS mean=least-squares mean; mITT=modified intent-to-treat; NT-proBNP=N-terminal pro–B-type natriuretic peptide; NYHA=New York Heart Association; RR=risk reduction; TTR=transthyretin.
References: 1. Attruby. Prescribing information. BridgeBio Pharma, Inc.; 2024. 2. Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2):132-142. doi:10.1056/NEJMoa2305434 3. Data on file. BridgeBio Pharma, Inc.; 2024-2025. 4. Judge DP, Heitner SB, Falk RH, et al. Transthyretin stabilization by AG10 in symptomatic transthyretin amyloid cardiomyopathy. J Am Coll Cardiol. 2019;74(3):285-295. doi:10.1016/j.jacc.2019.03.012 5. Maurer MS, Judge DP, Gillmore JD, et al. Early increase in serum transthyretin by acoramidis independently predicts improved survival in TTR amyloid cardiomyopathy. J Am Coll Cardiol. 2025;85 (20): 1911-1923. doi:10.1016/j.jacc.2025.03.542 6. Sarswat N, Ambardekar A, Taubel J, et al. Acoramidis-mediated early increase in serum transthyretin is associated with lower cardiovascular-related hospitalizations and mortality: insights from the ATTRibute-CM study. Poster presented at: American College of Cardiology Annual Scientific Meeting; March 29-31, 2025; Chicago, IL. 7. Maurer MS, Masri A, Cappelli F, et al. Increase in serum TTR levels observed with acoramidis treatment in patients with transthyretin amyloid cardiomyopathy (ATTR-CM): insights from ATTRibute-CM and its open-label extension. Poster presented at: European Society of Cardiology Congress; August 30-September 2, 2024; London, UK.