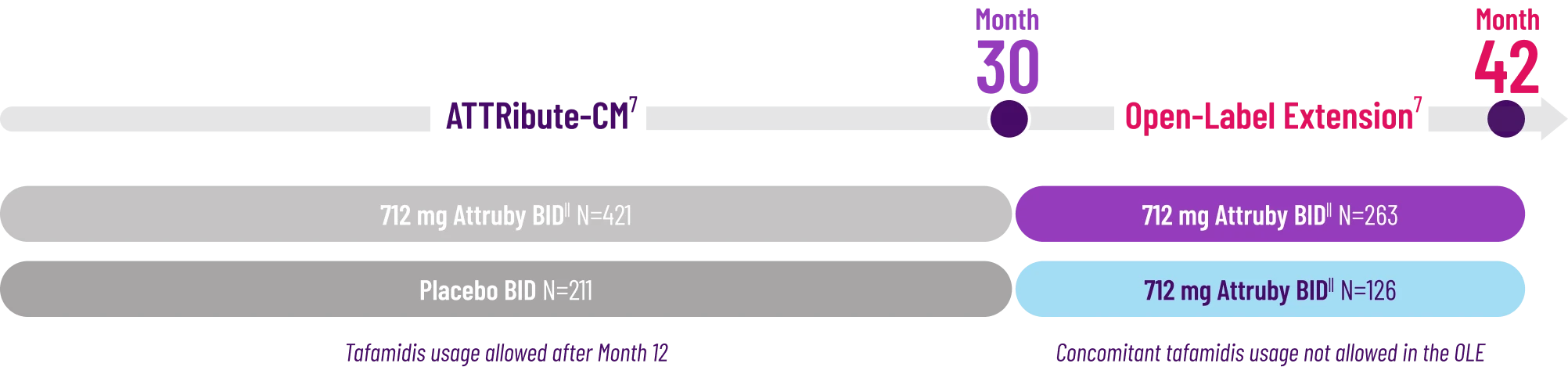It starts with what's
at stake. And determines
what comes next.
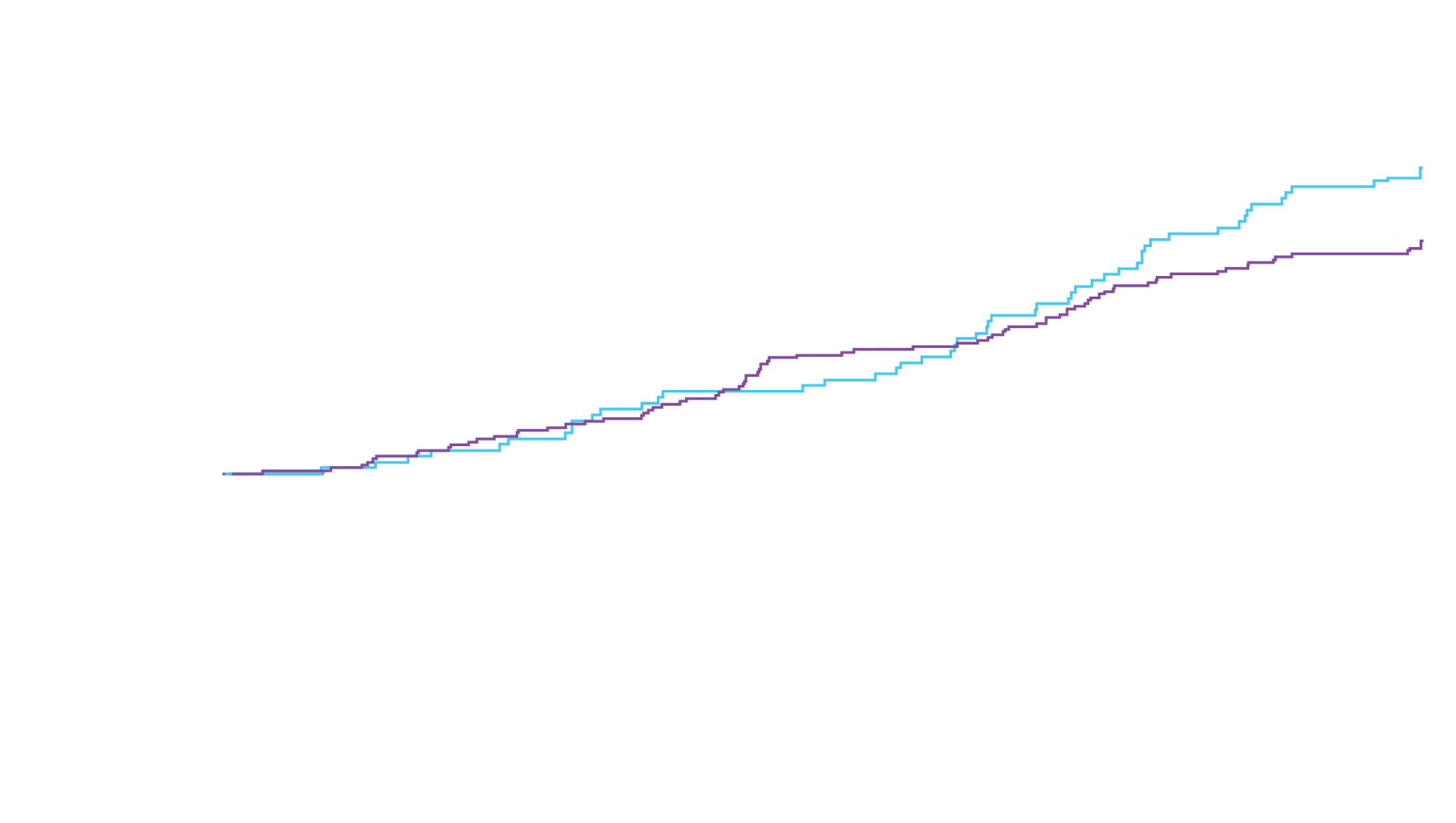
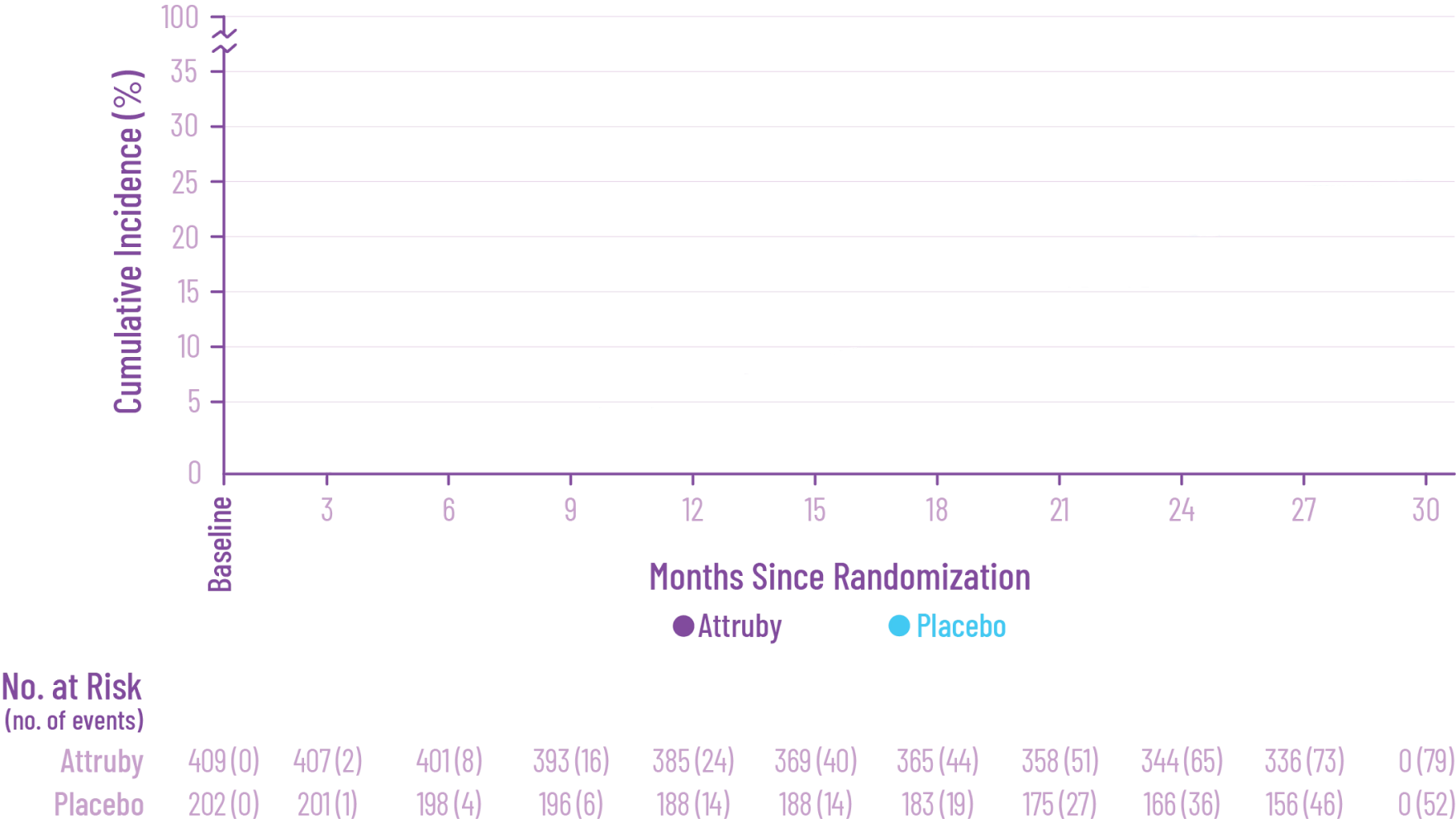
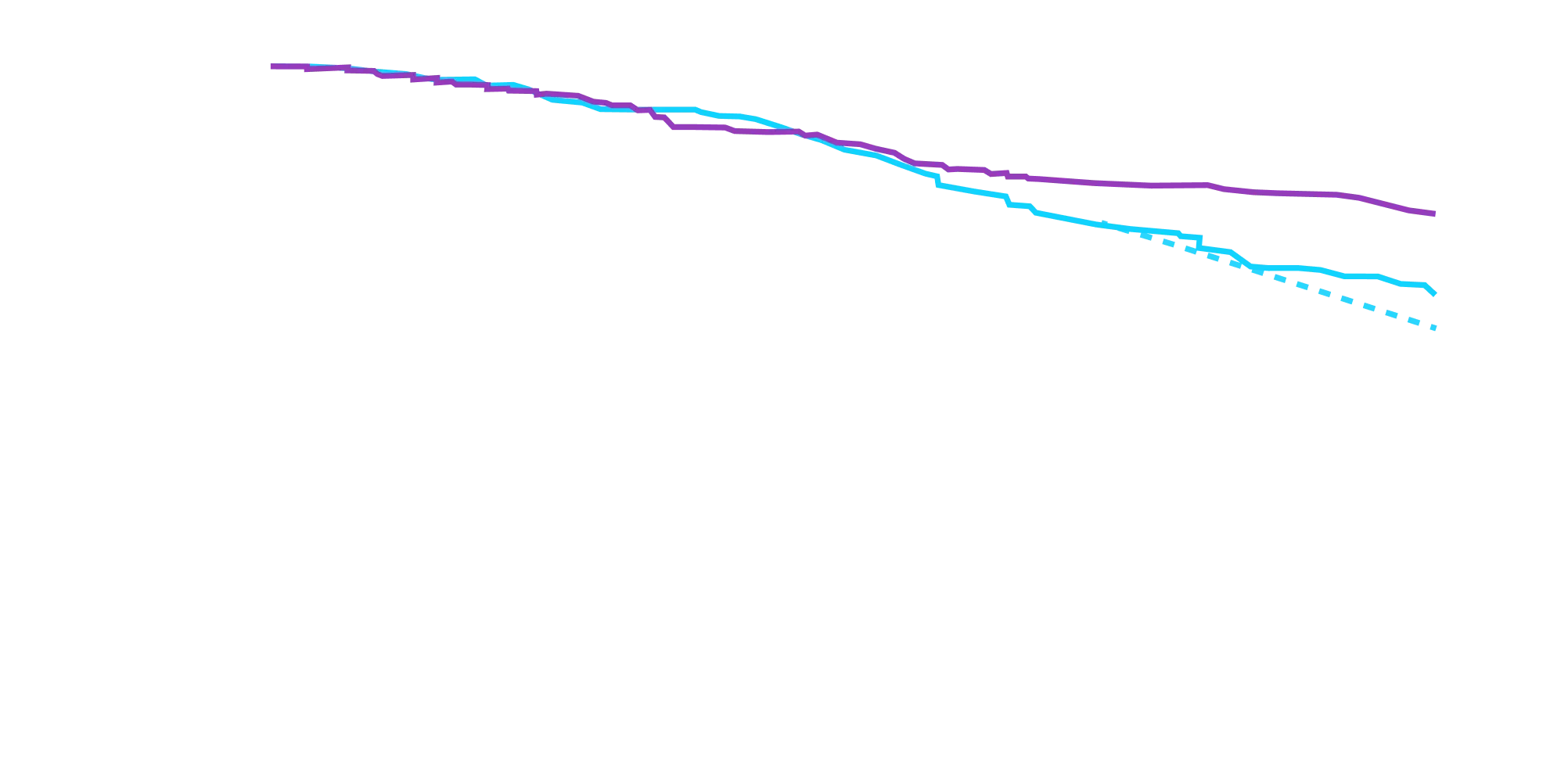
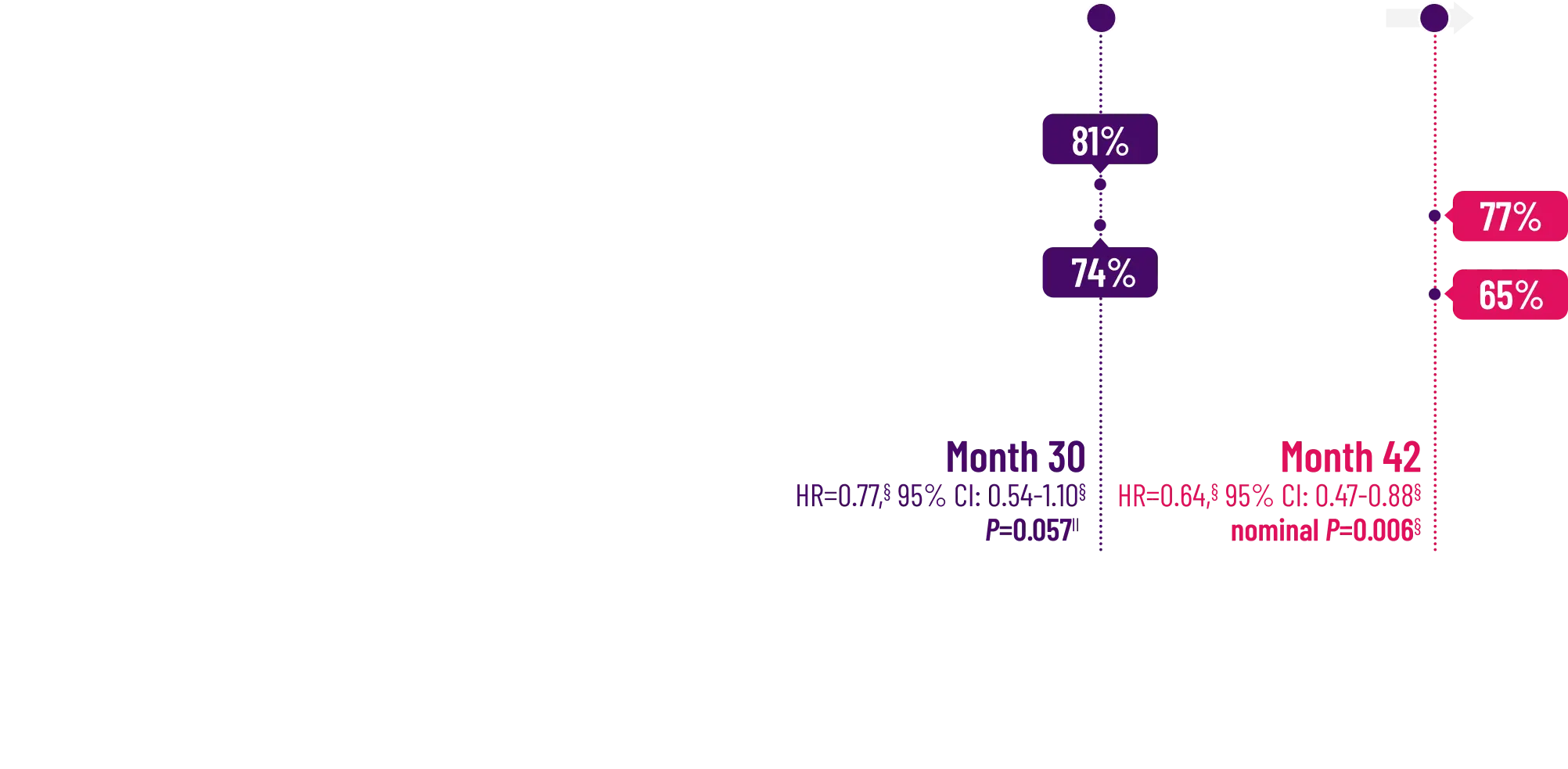
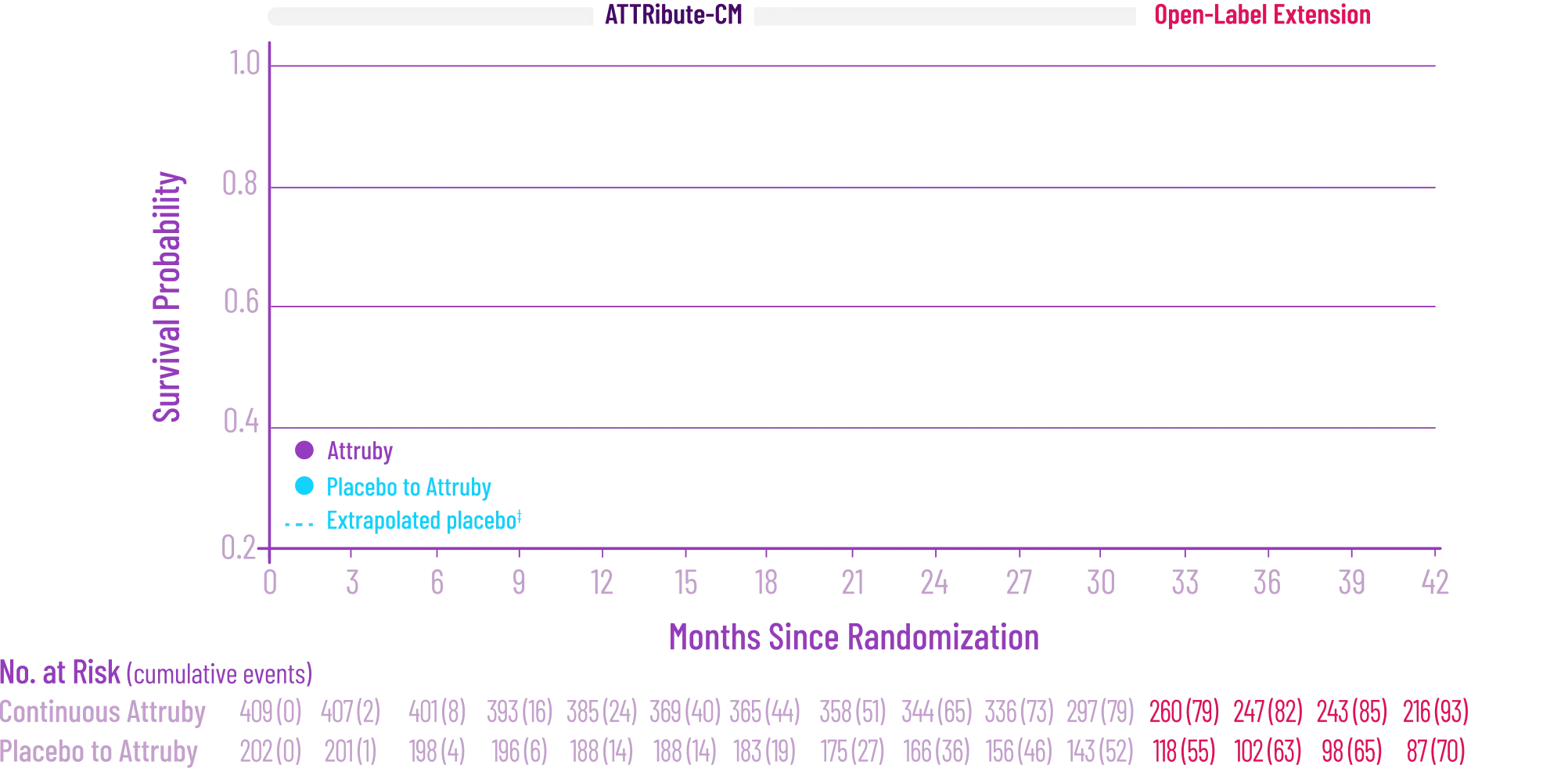
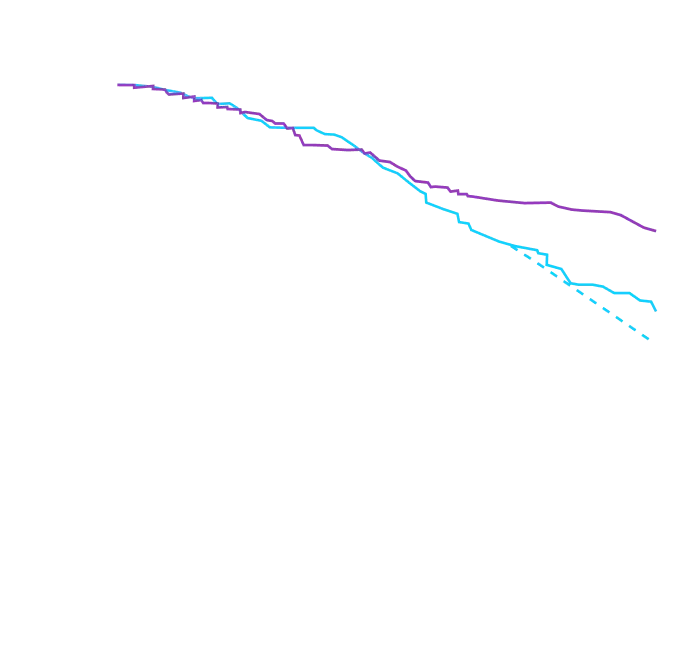
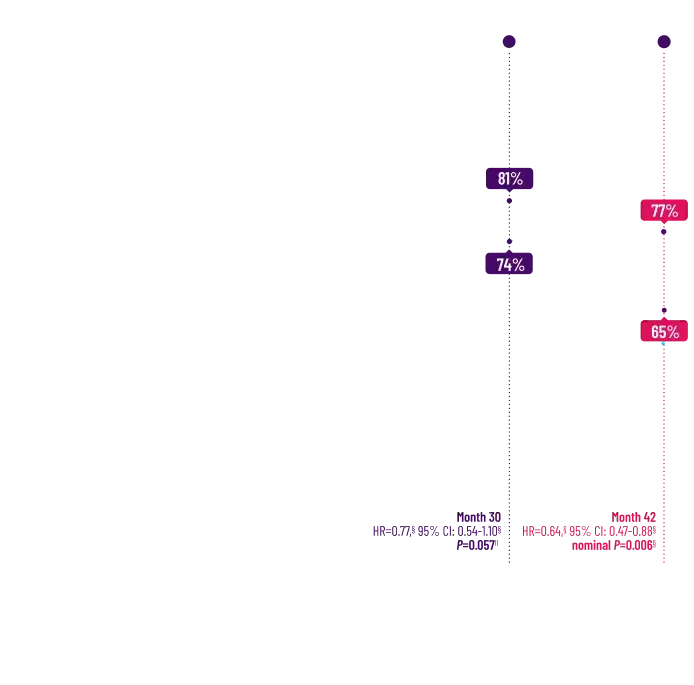
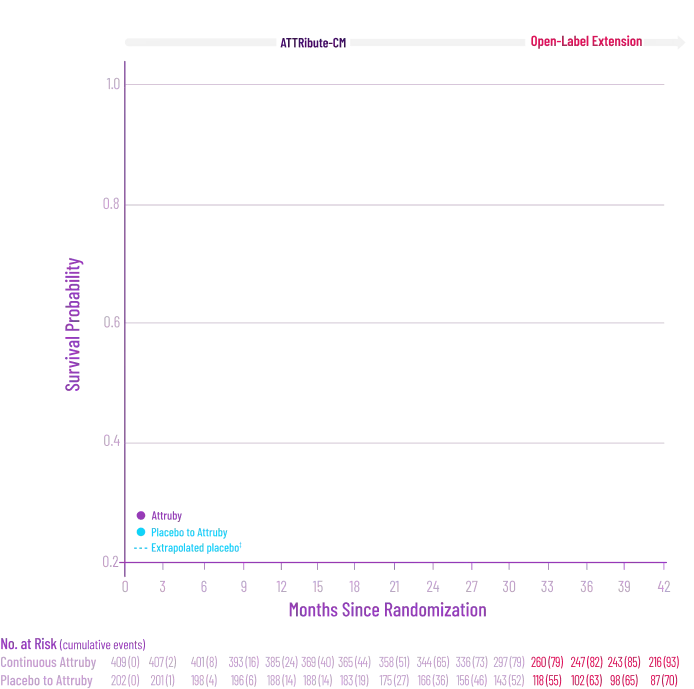
remained alive at 30 months
vs 74% with placebo1
CMH P=0.057.3
All-Cause Mortality (ACM)2
The treatment effect of Attruby on ACM was studied in a contemporary ATTR-CM population that reflects recent approaches in diagnosis and care2,4†
In a post hoc analysis applied to the ITT‡ population, which included patients with stage 4 CKD,
Attruby reduced the risk of ACM5
Cox HR=0.762 (95% CI: 0.524-1.072);
CMH P=0.0396
†The proportion of trial participants in the earlier stages of disease (ie, NYHA Class I and II)
reflects changes in disease recognition and management in recent
years, which have resulted in improved overall survival.2,4
‡Based on ITT population of 632 patients, including 21 who had stage 4 CKD. The ITT population was defined
as all randomized participants who received at least
1 dose of study drug and had at least 1 postbaseline efficacy evaluation and
included participants who had a baseline eGFR <30 mL/min/1.73 m2.2
ACM=all-cause mortality; CKD=chronic kidney disease; CMH=Cochran-Mantel-Haenzel test; CVM=cardiovascular-related mortality; eGFR=estimated glomerular filtration rate; HR=hazard ratio; ITT=intent-to-treat; NYHA=New York Heart Association.
References: 1. Attruby. Prescribing information. BridgeBio Pharma, Inc.; 2024. 2. Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2):132-142. doi:10.1056/NEJMoa2305434 3. Data on file. BridgeBio, Inc.; 2024-2025. 4. Ioannou A, Patel RK, Razvi Y, et al. Impact of earlier diagnosis in cardiac ATTR amyloidosis over the course of 20 years. Circulation. 2022;146(22):1657-1670. doi:10.1161/CIRCULATIONAHA.122.060852 5. Poulsen S, Gillmore JD, Alexander KM, et al. ATTRibute-CM: ITT sensitivity analysis and sub-analysis comparing acoramidis and placebo in stage 4 CKD. Paper presented at: 2024 International Symposium on Amyloidosis; May 26-30, 2024; Rochester, MN. 6. Judge DP, Gillmore JD, Alexander KM, et al. long-term efficacy and safety of acoramidis in ATTR-CM: initial report from the open-label extension of the ATTRibute-CM trial. Circulation. 2025;151(9): 601-611. doi:10.1161/CIRCULATIONAHA.124.072771 7. Judge DP, Gillmore JD, Alexander KM, et al. Long-term efficacy and safety of acoramidis in ATTR-CM: initial report from the open-label extension of the ATTRibute-CM trial. Circulation. 2025;151(suppl 1):1-4. doi:10.1161/CIRCULATIONAHA.124.072771