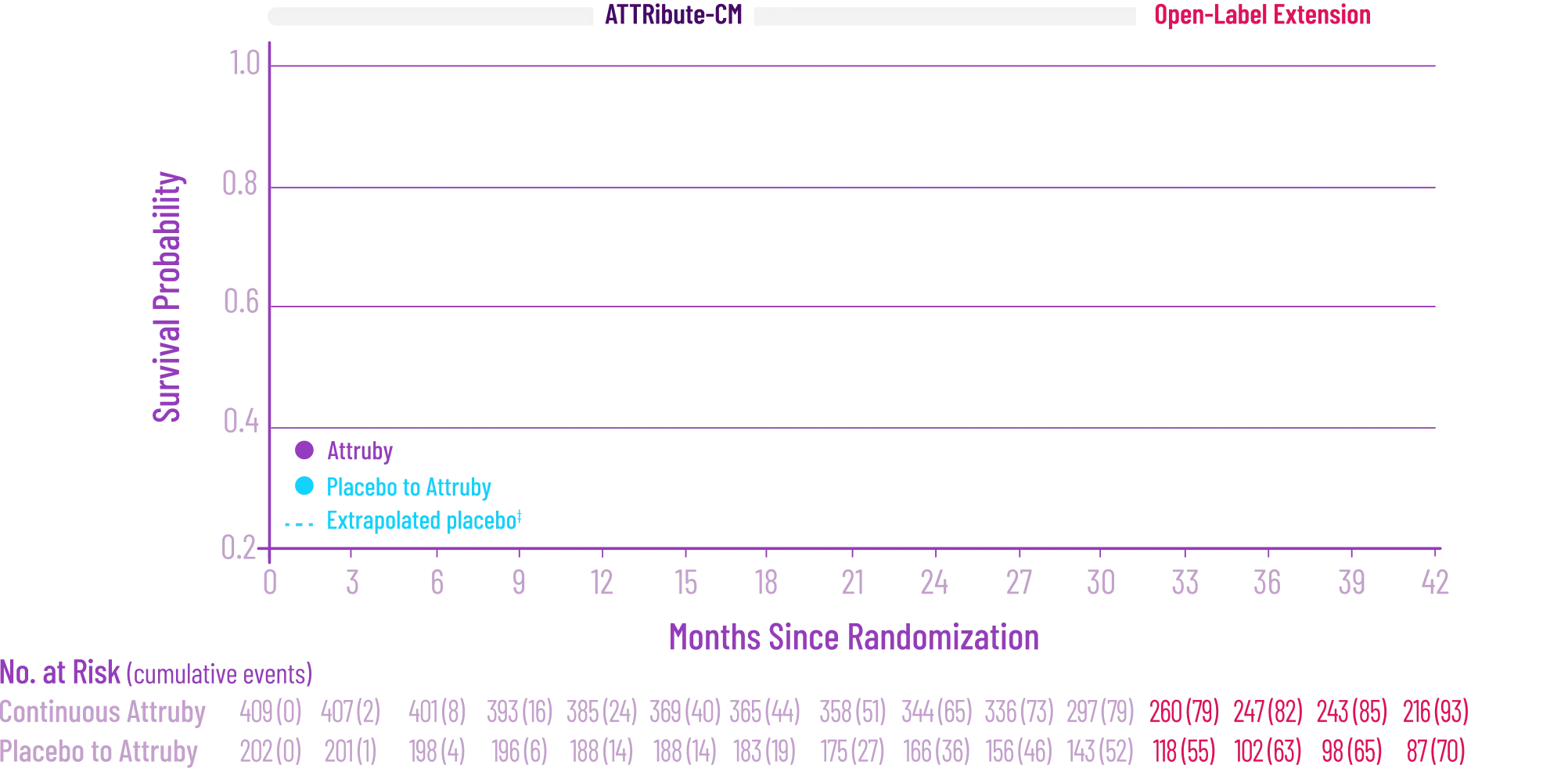
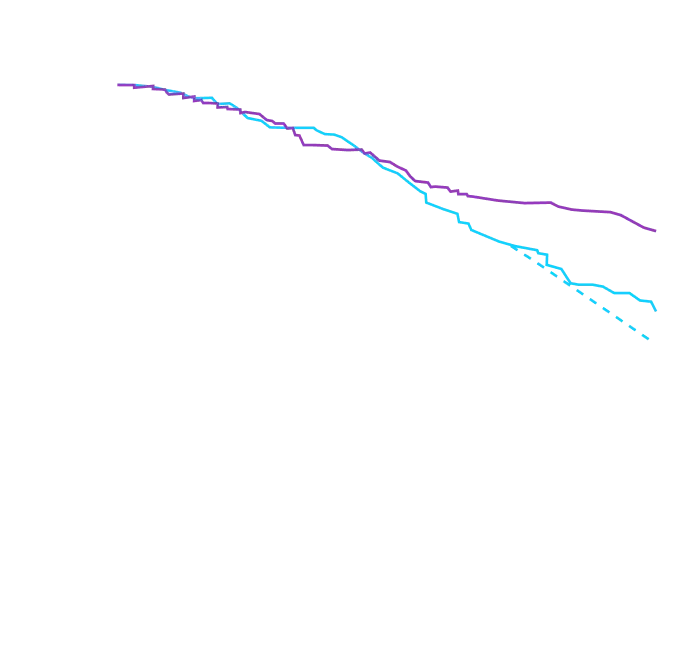
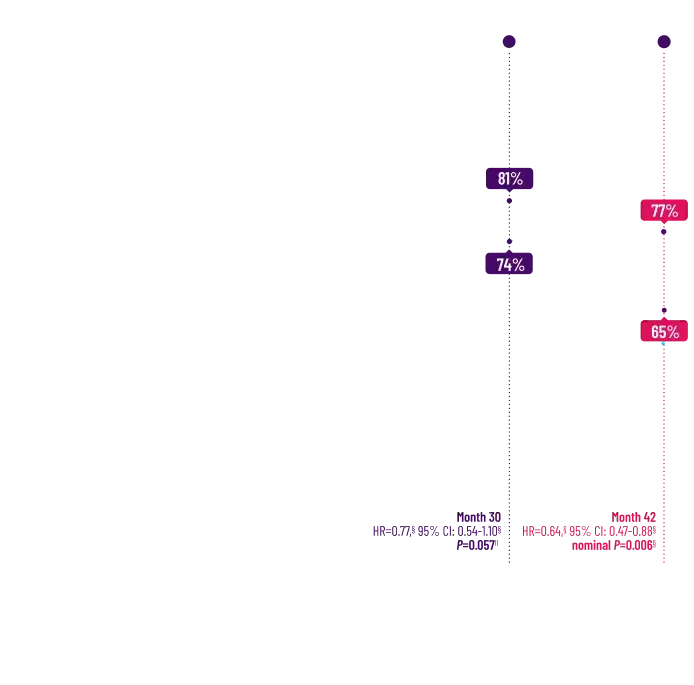
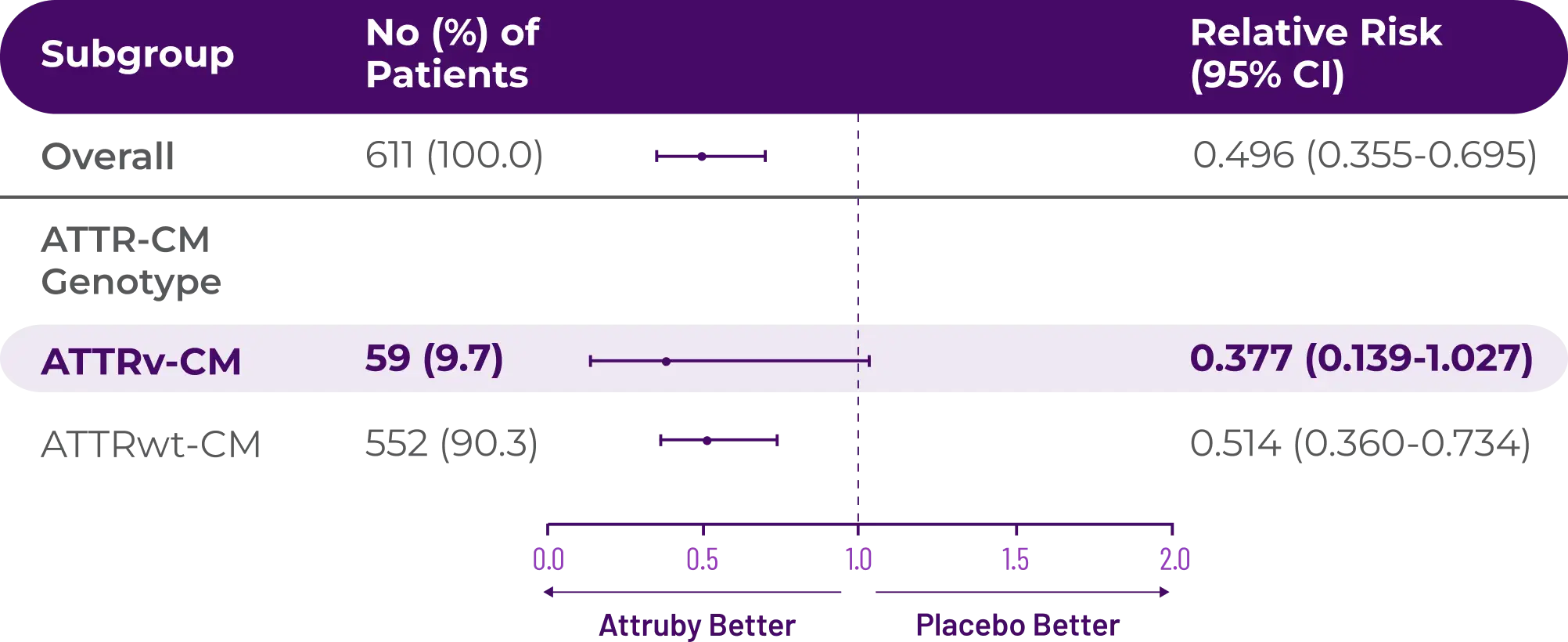
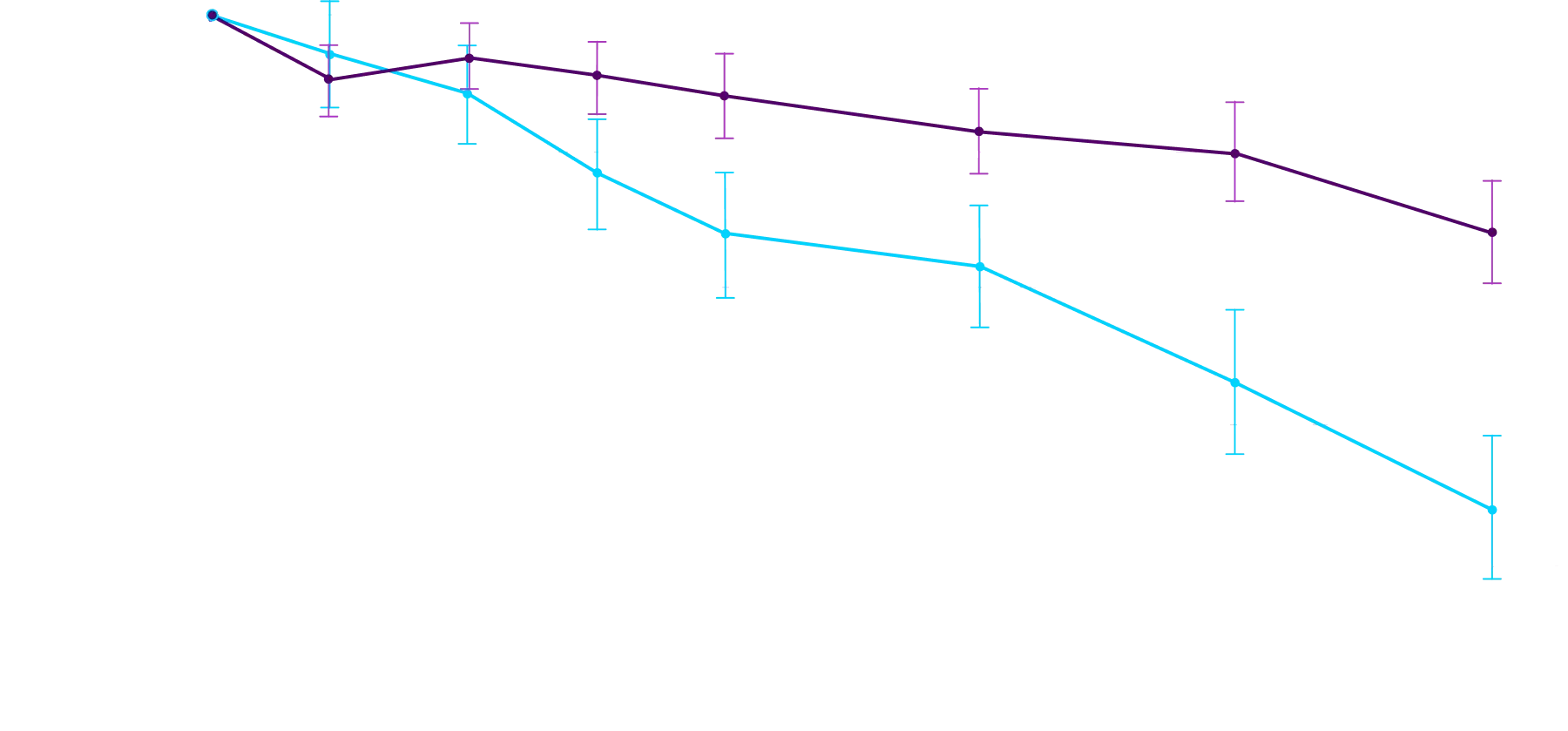
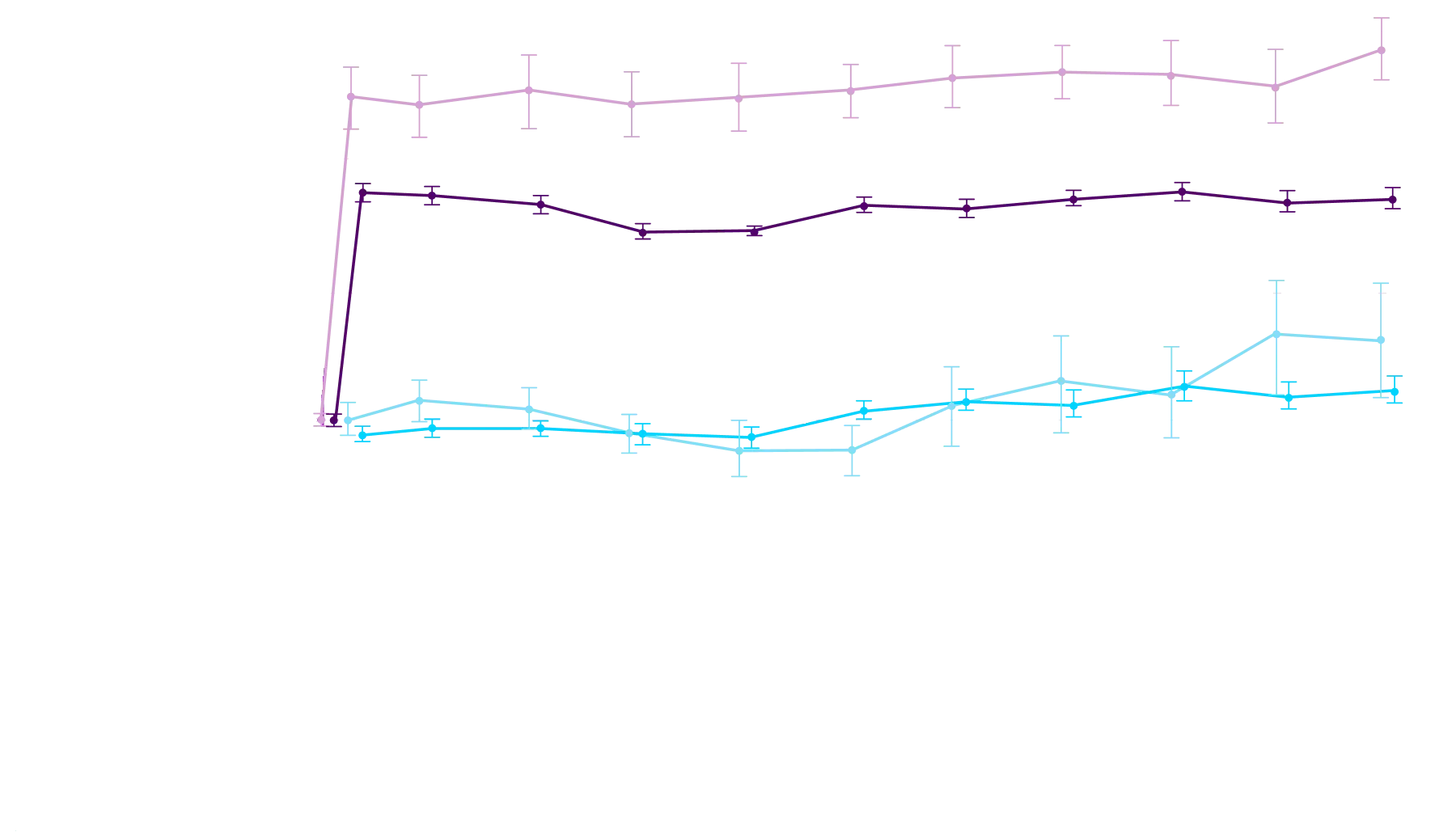
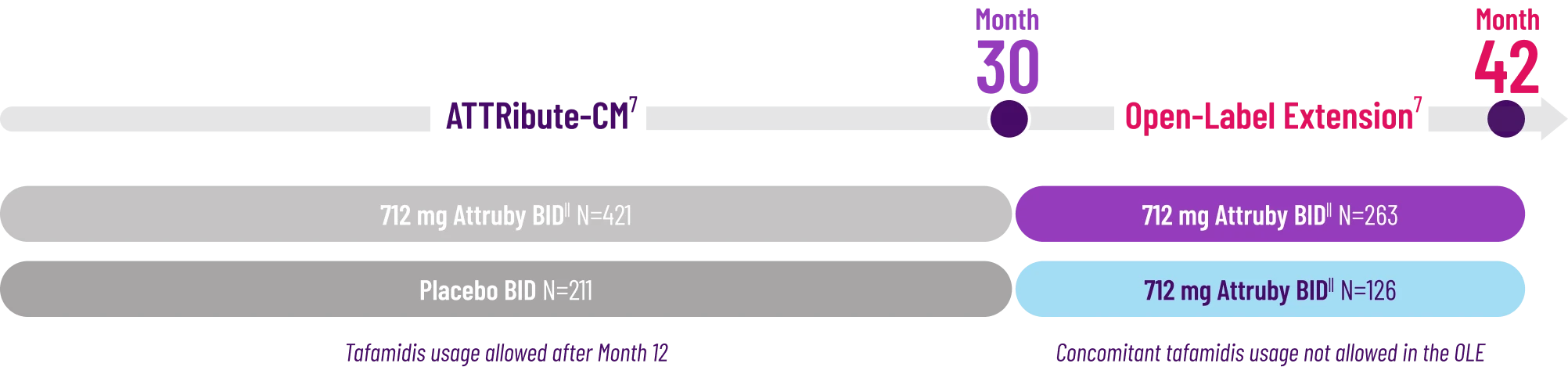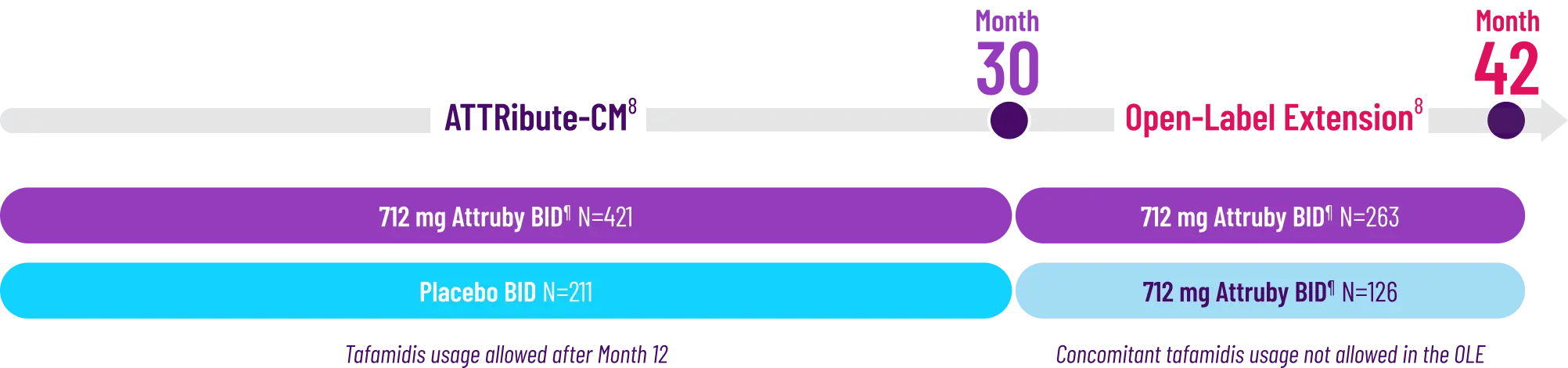Attruby made an impact
on NT-proBNP levels
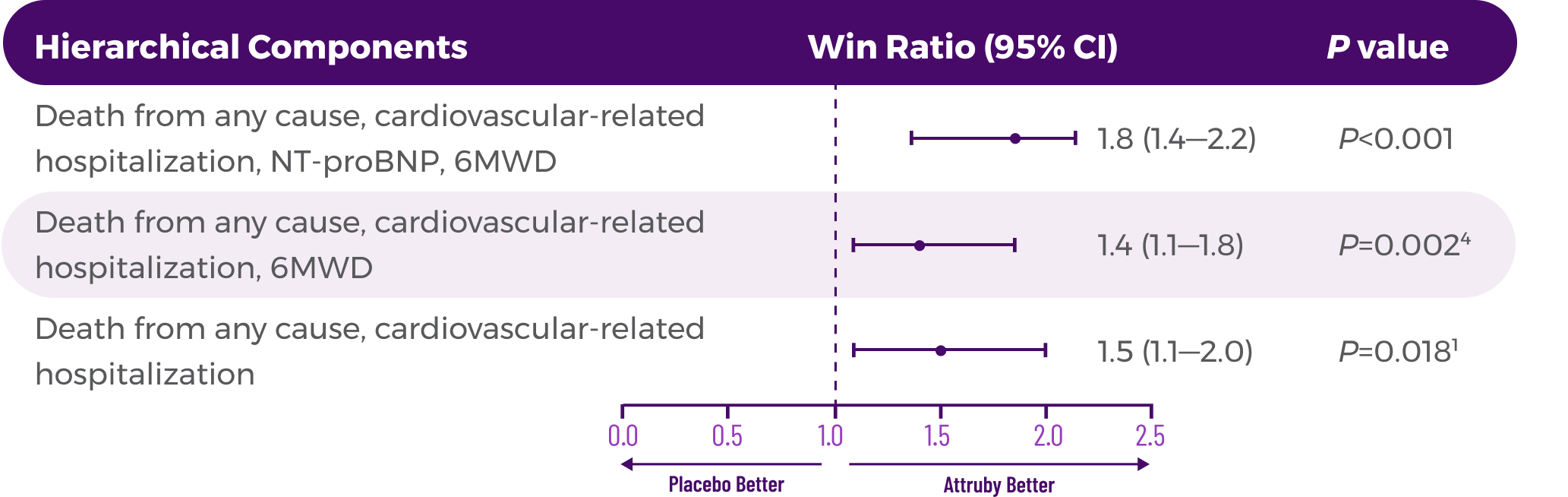
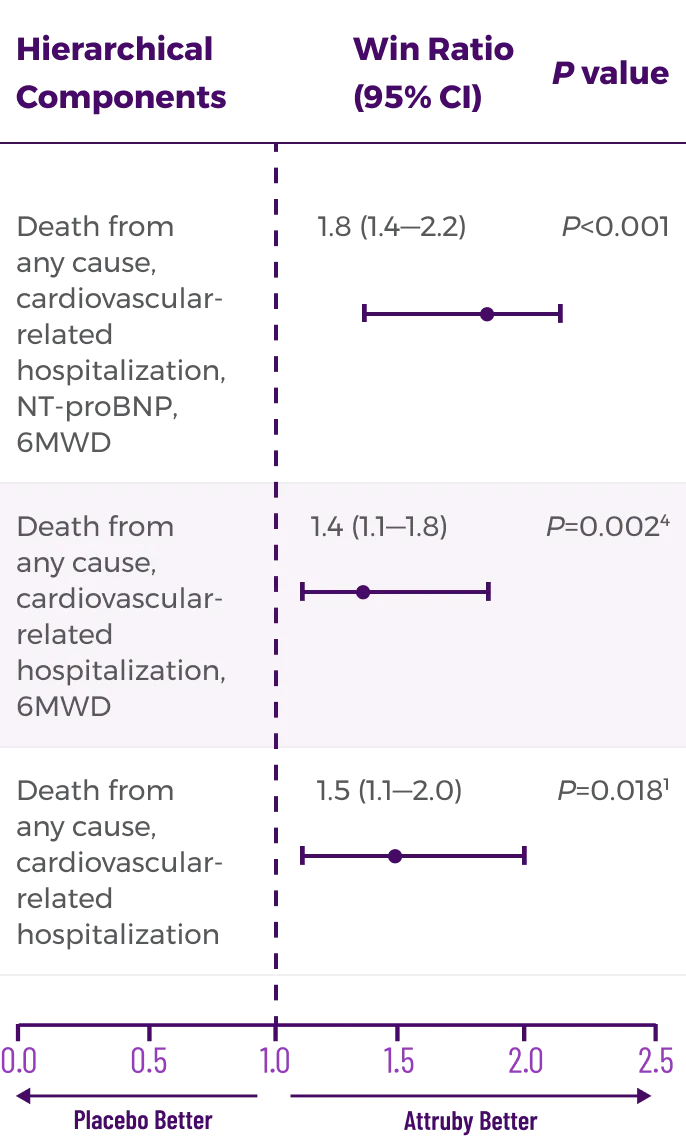
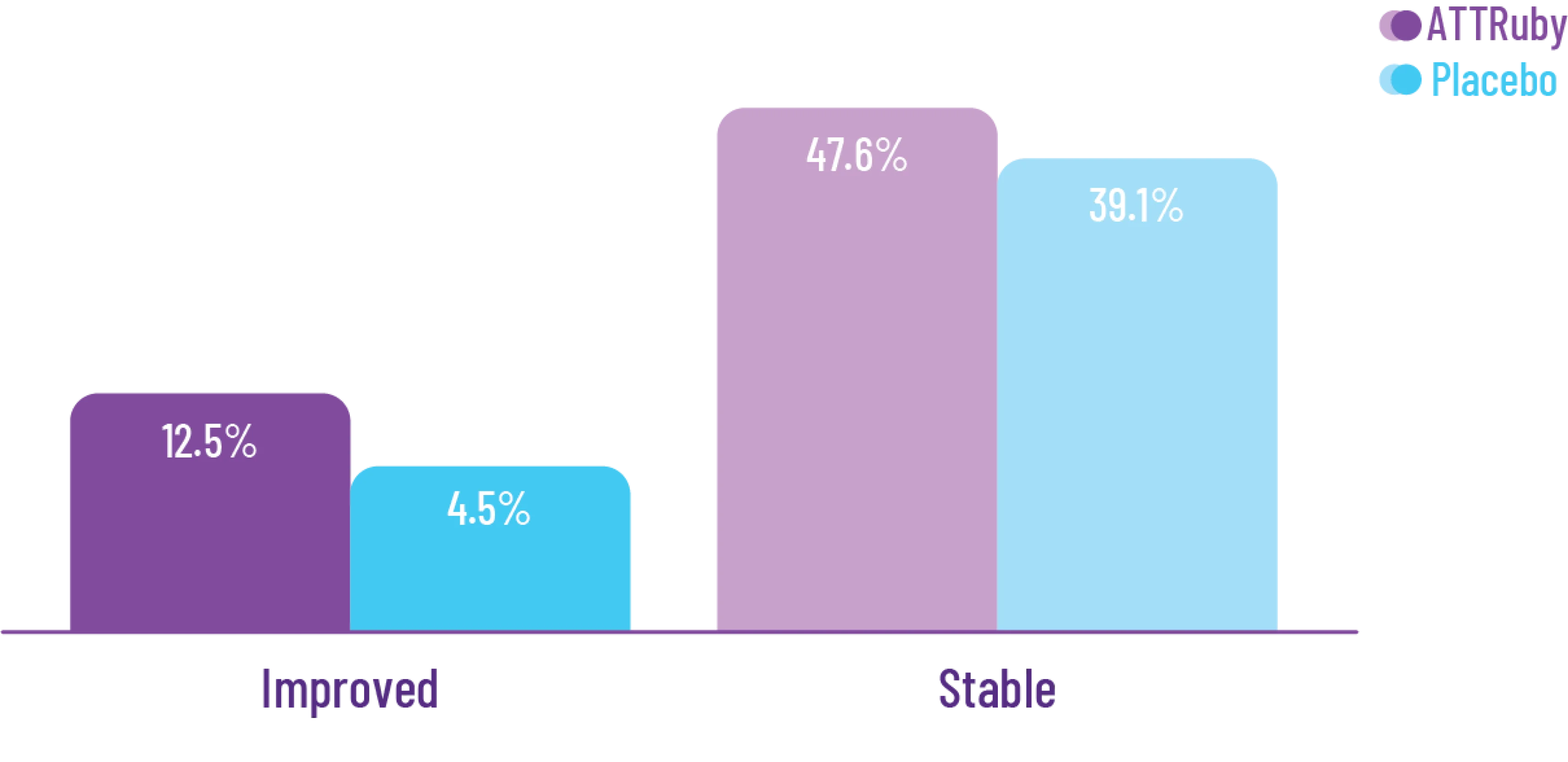
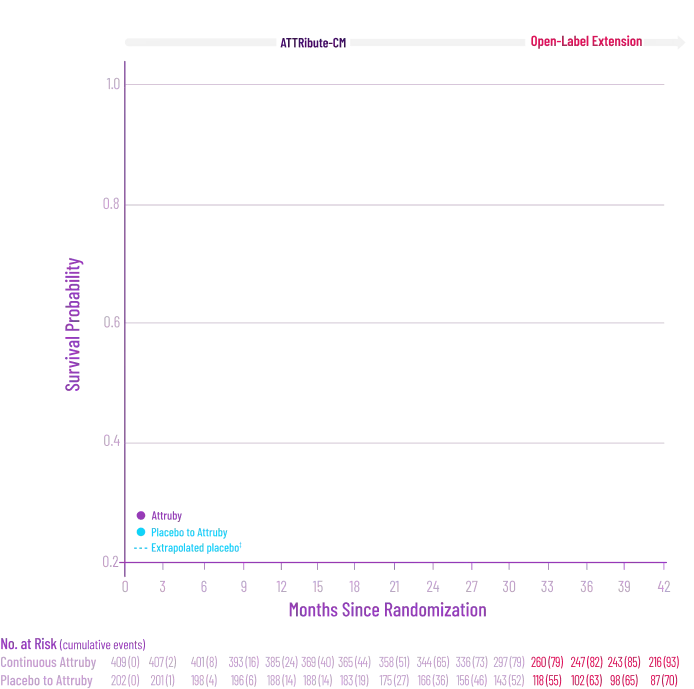
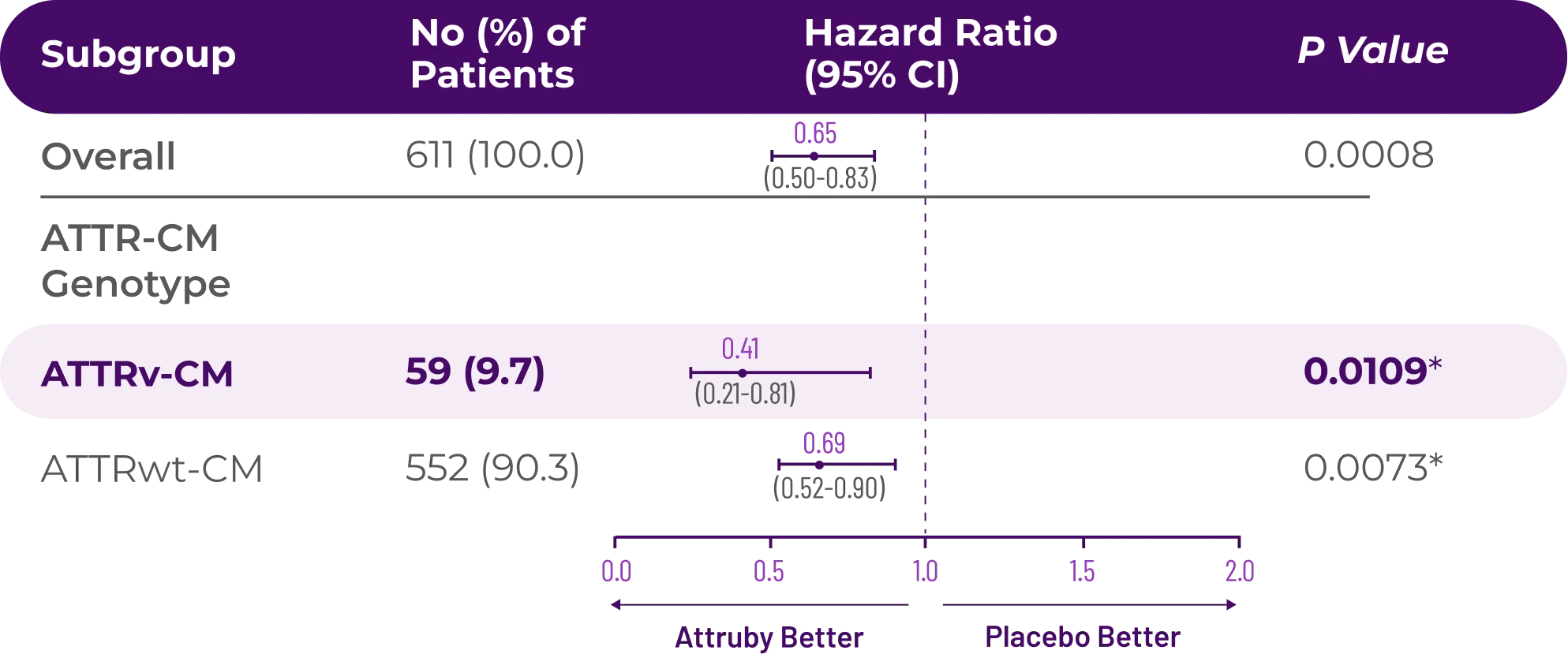
Other Secondary Endpoint:
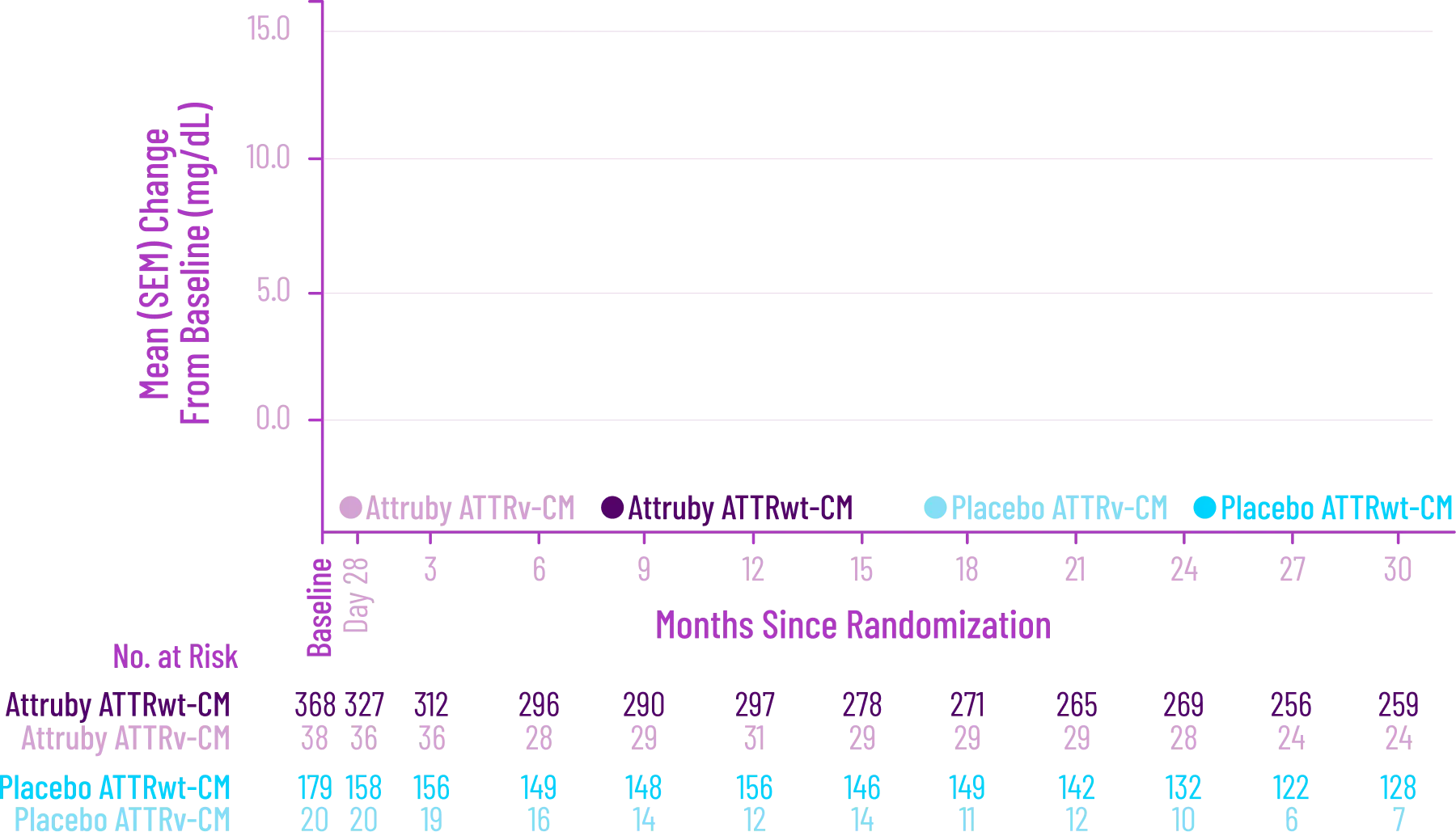
Change From Baseline in NT-proBNP
Through Month 302†
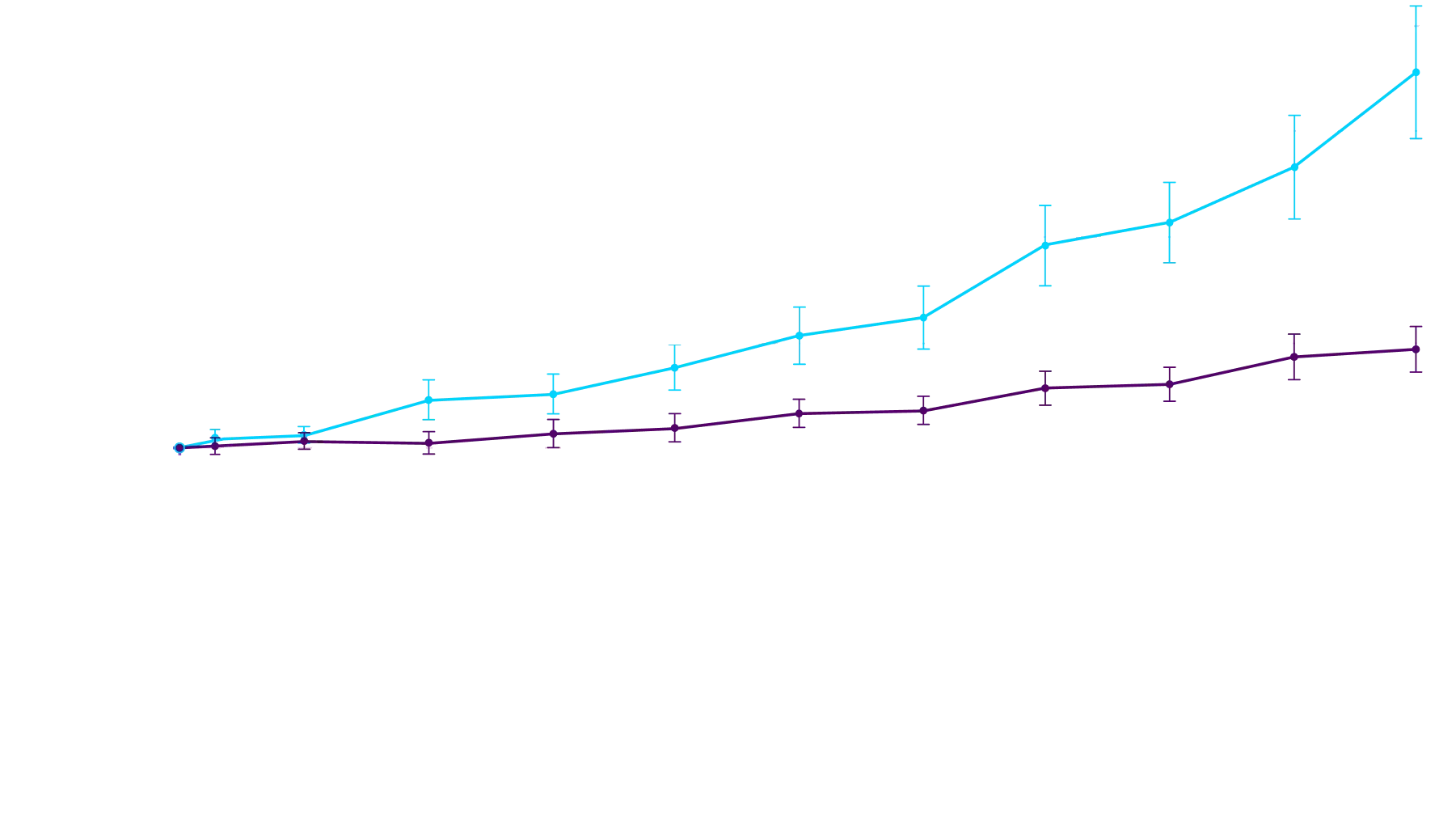
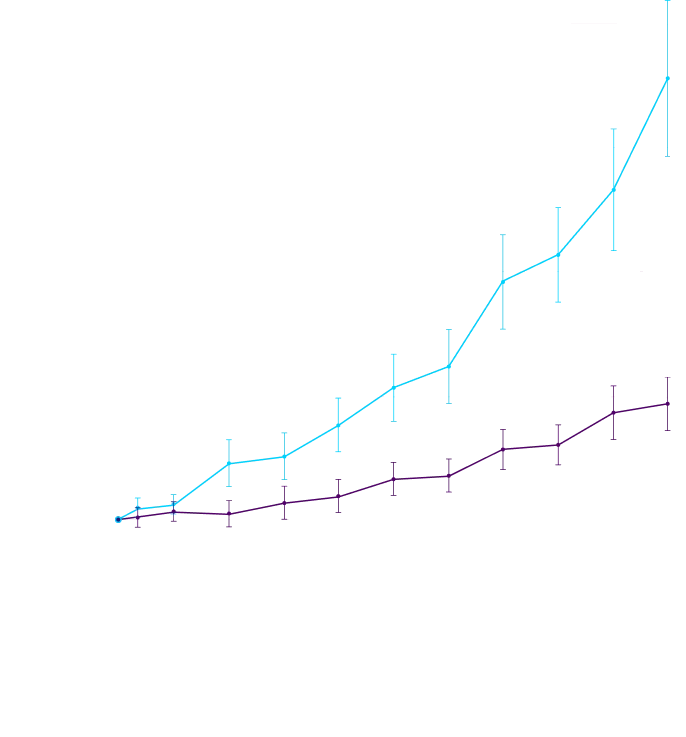


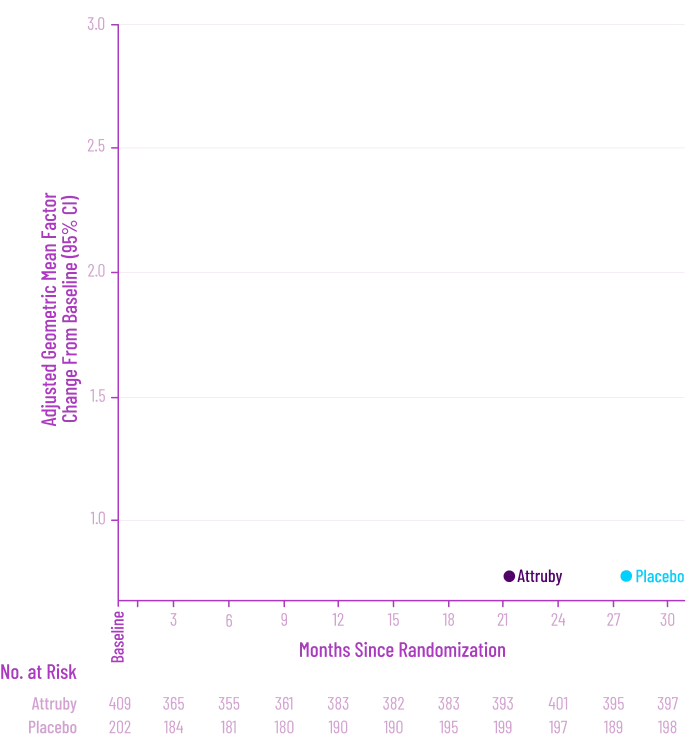
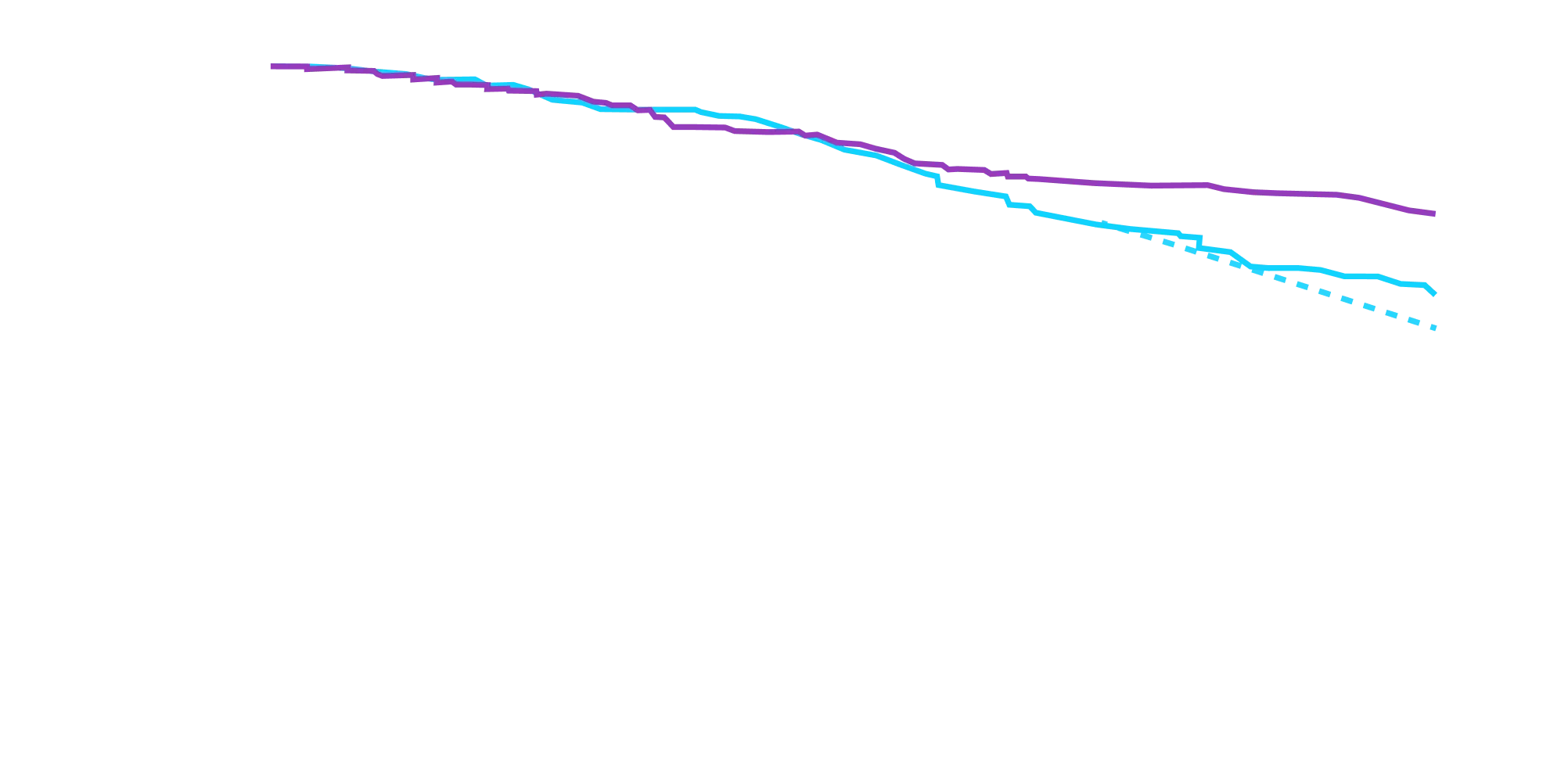
 CHANGE IN NT-proBNP
CHANGE IN NT-proBNPFAVORED ATTRUBY
Elevated levels of NT-proBNP are an indicator of heart stress3
†Prespecified, non-alpha protected secondary endpoint.4
NT-proBNP=N-terminal pro–B-type natriuretic peptide.
References: 1. Attruby. Prescribing information. BridgeBio Pharma, Inc.; 2024.
2. Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2):132-142. doi:10.1056/NEJMoa2305434
3. Suzuki S, Sugiyama S. The molar ratio of N-terminal
pro-B-type natriuretic peptide/B-type natriuretic peptide for heart failure-related events in stable outpatients with cardiovascular risk factors. Internal Medicine. 2018;57(18):2621-2630. doi:10.2169/internalmedicine.0471-17
4. Data on file. BridgeBio Pharma, Inc.; 2024-2025.