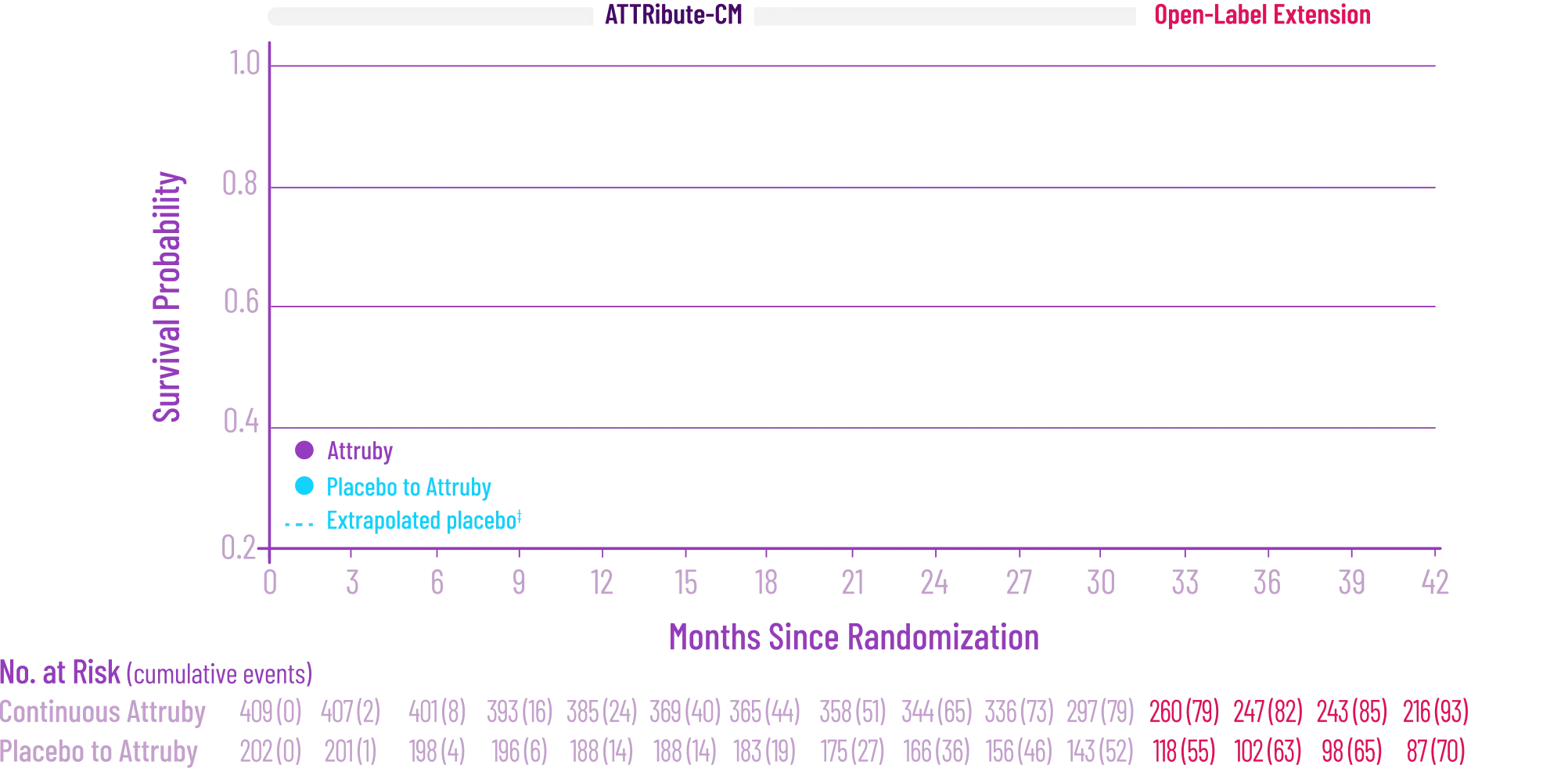
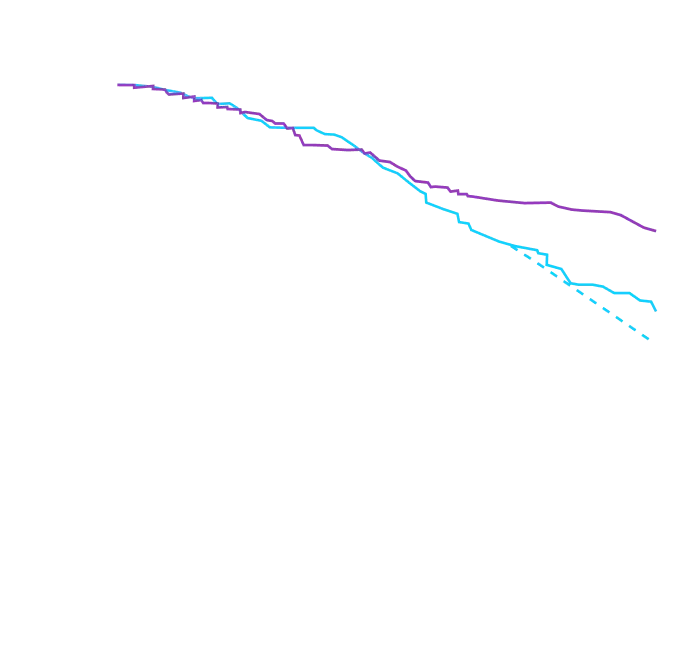
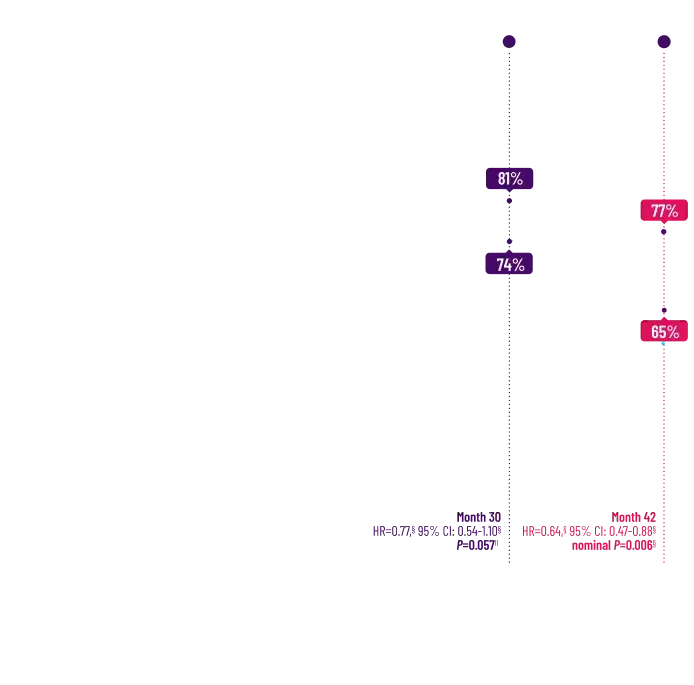
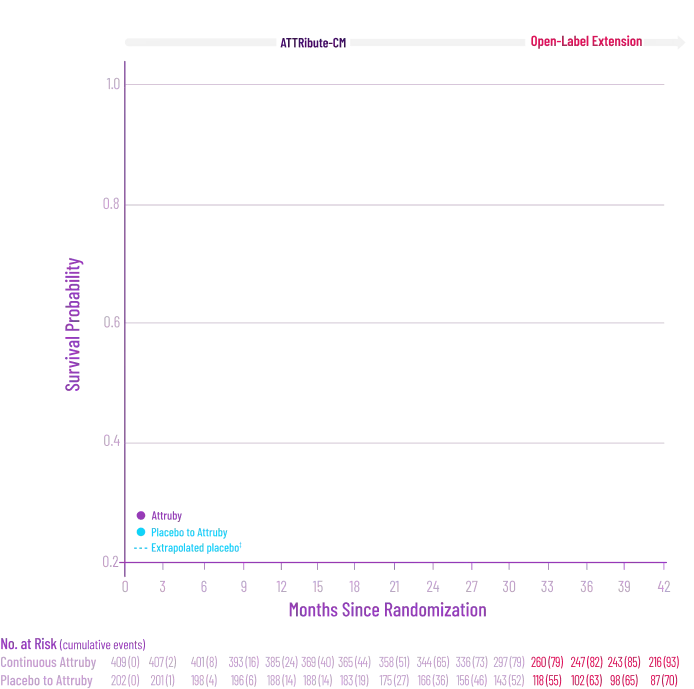
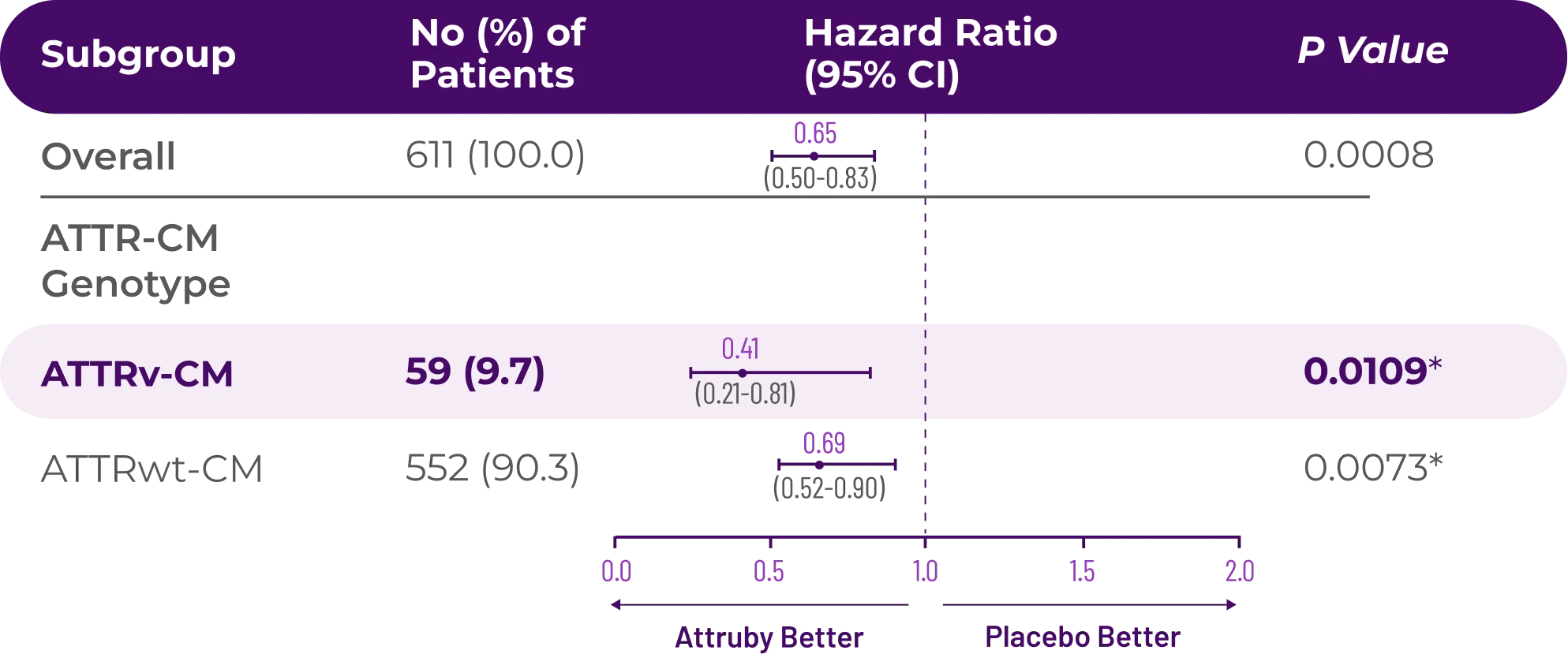
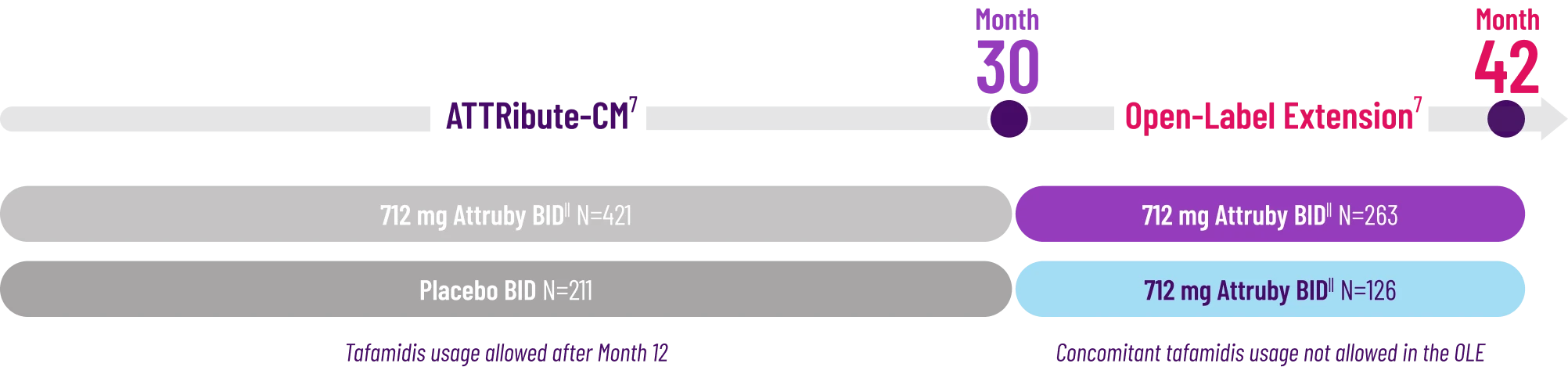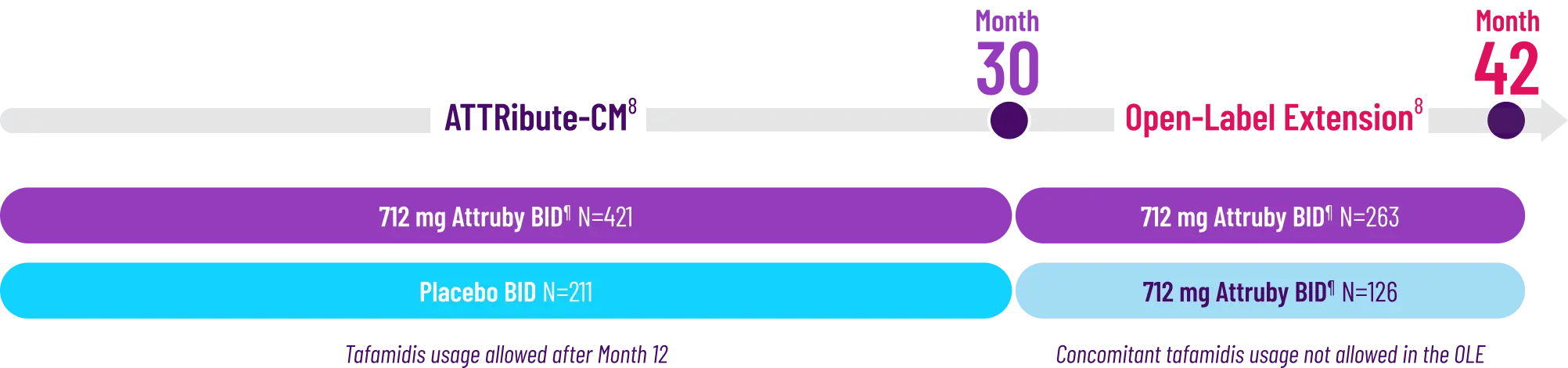It's more than data. It's how they feel over time.
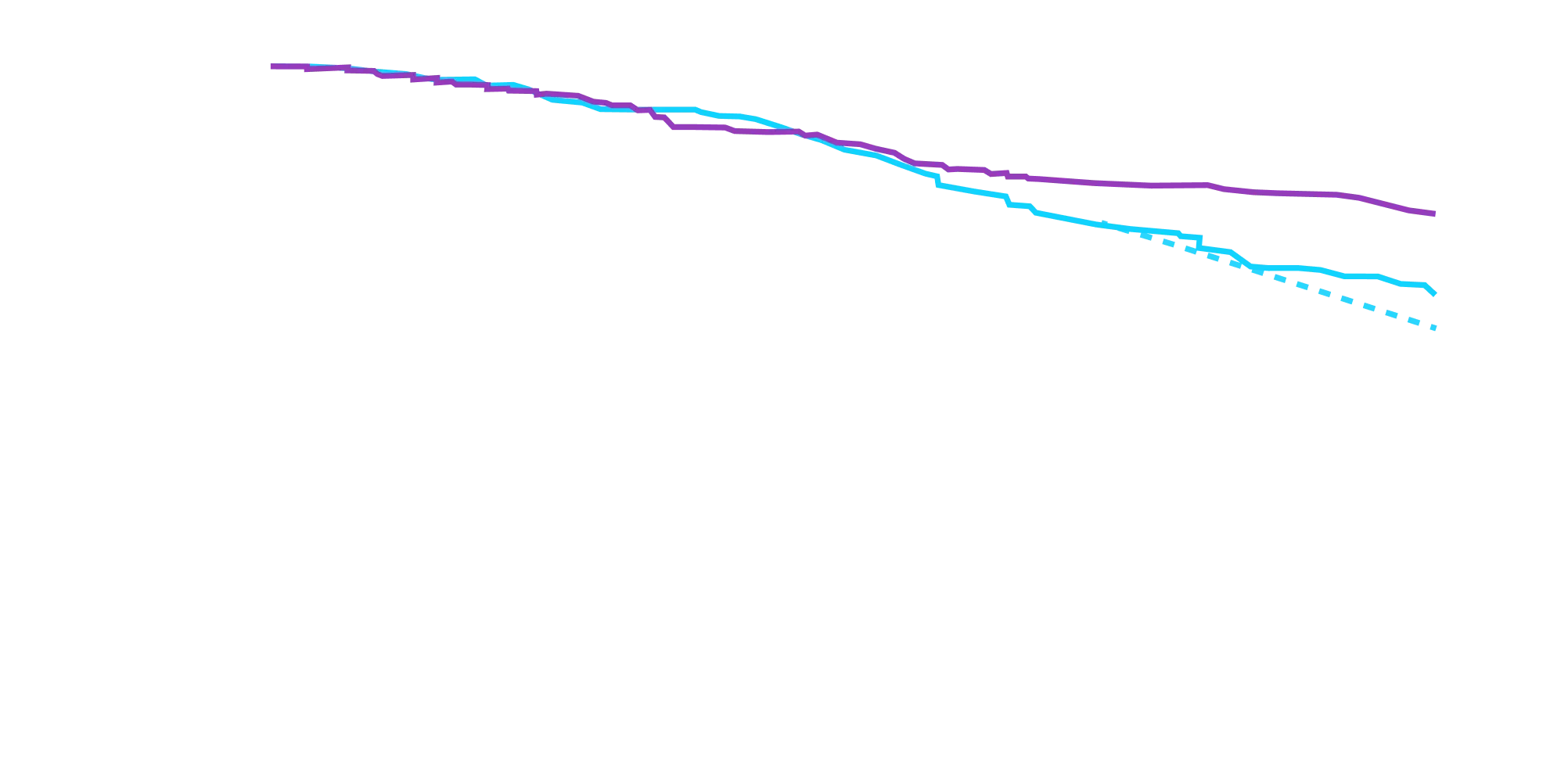
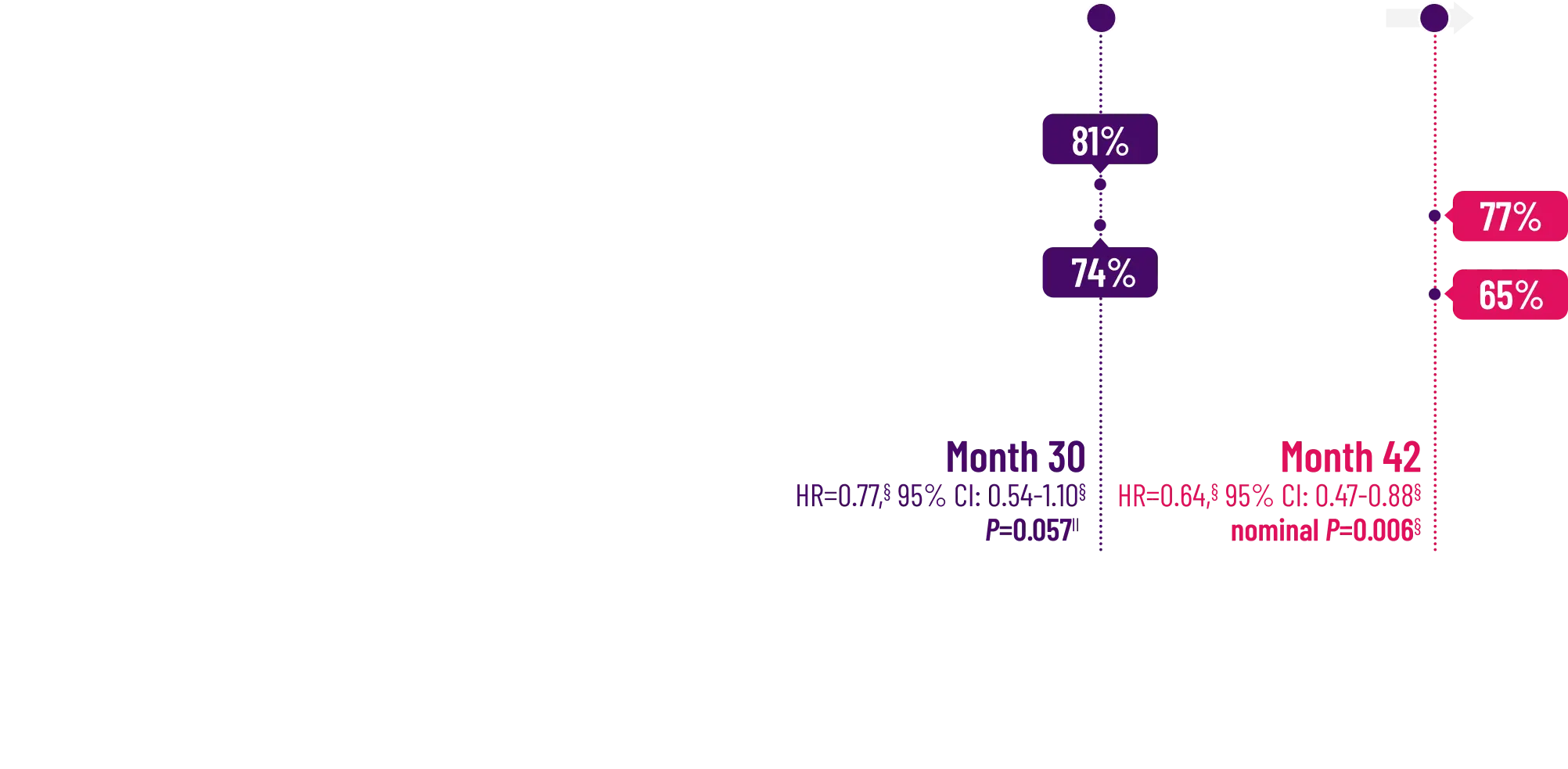
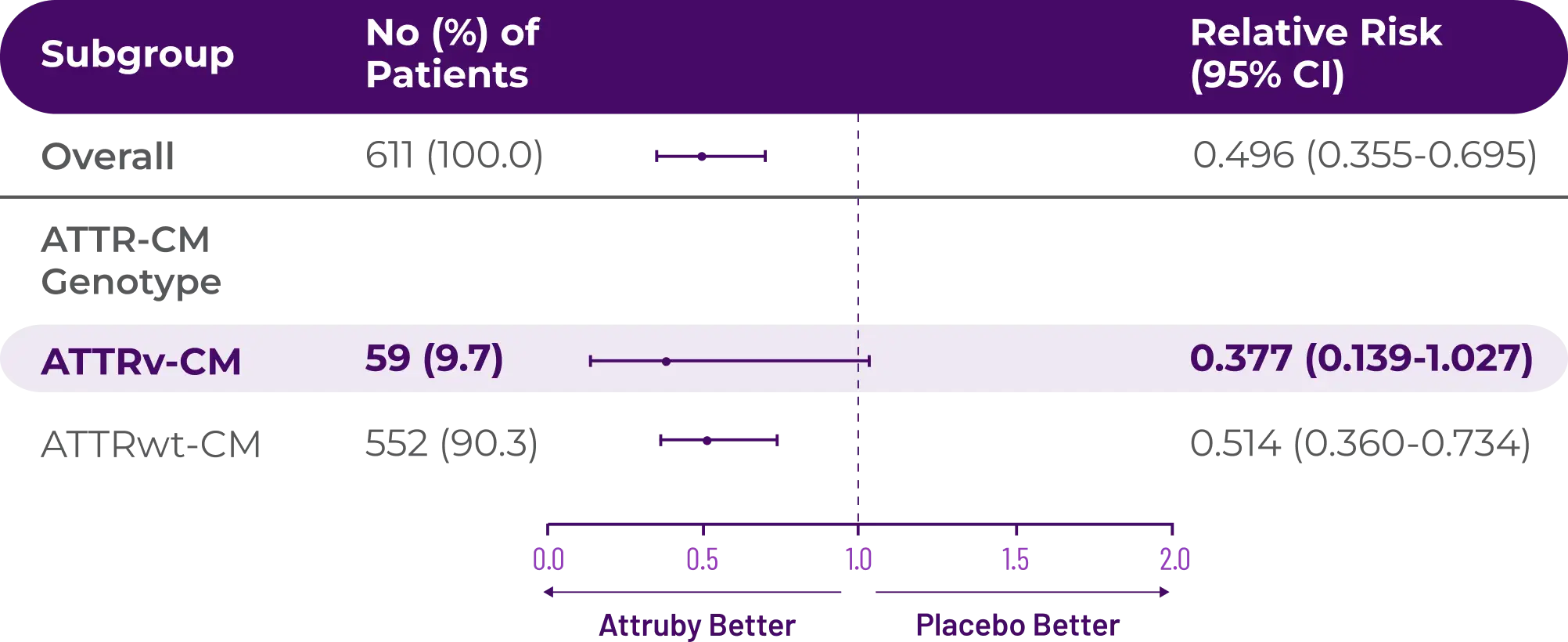
Attruby made a difference in health-related quality of life compared with placebo at 30 months1,2
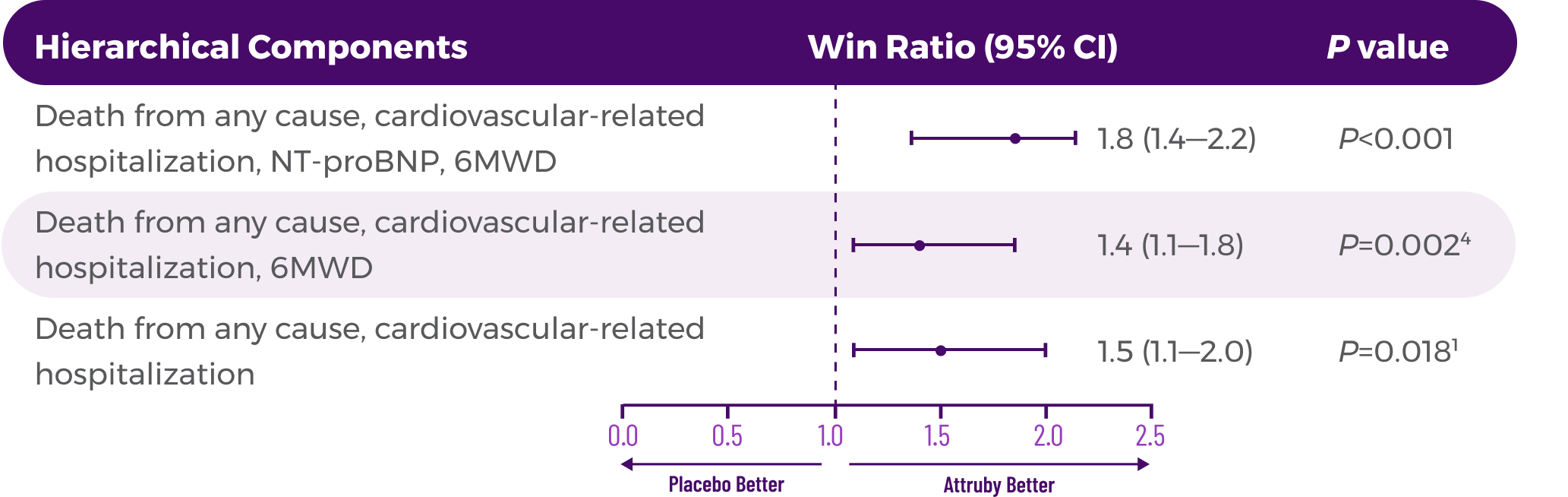
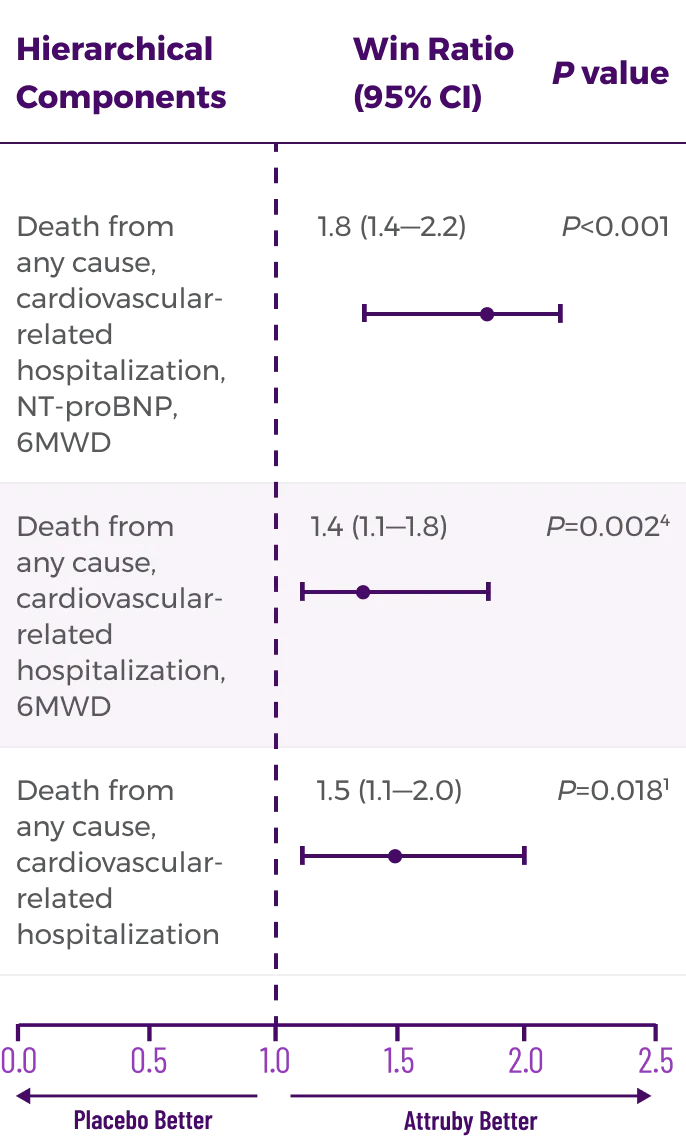
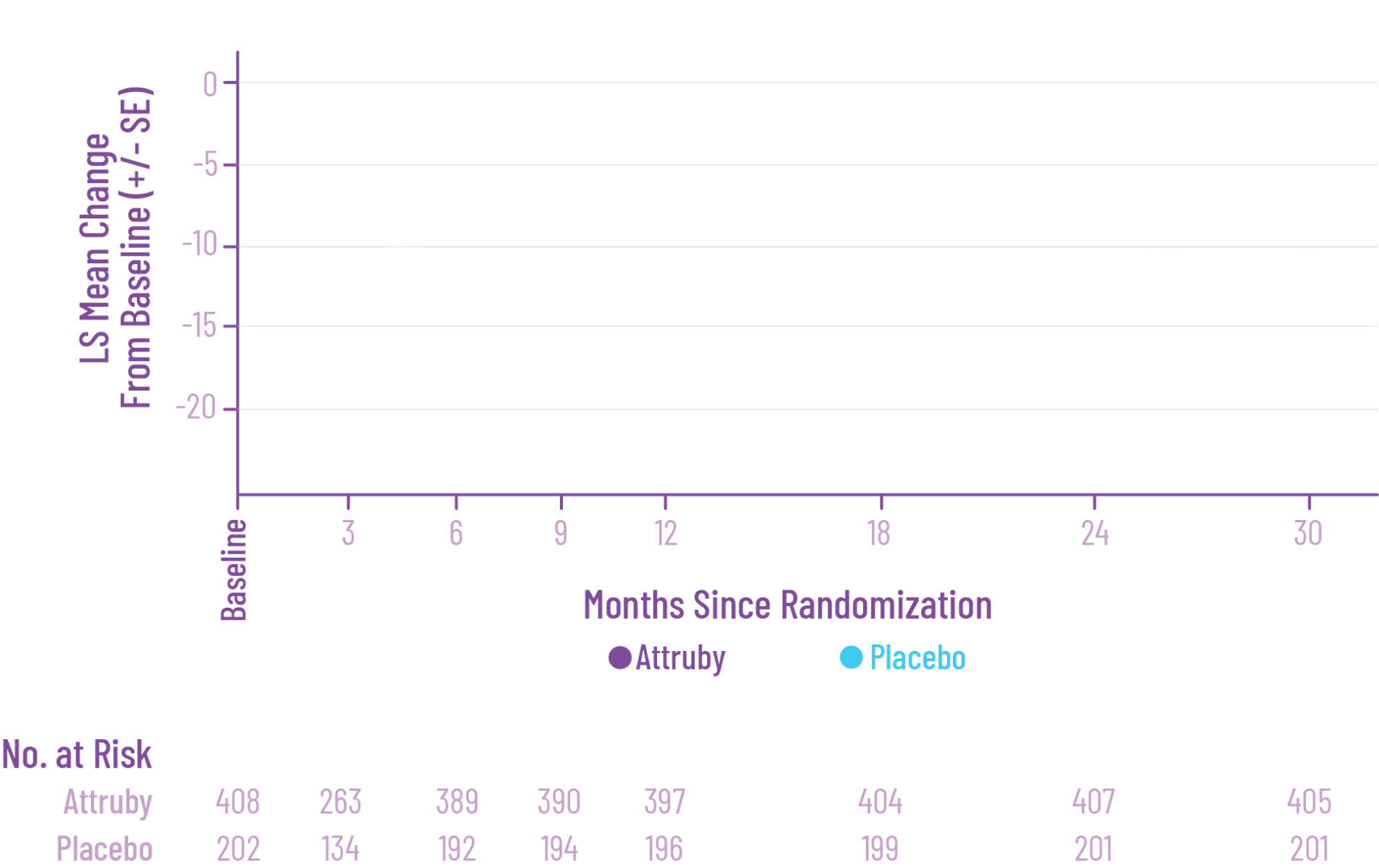
Key Secondary Endpoint:
Change From Baseline in KCCQ-OS Score1
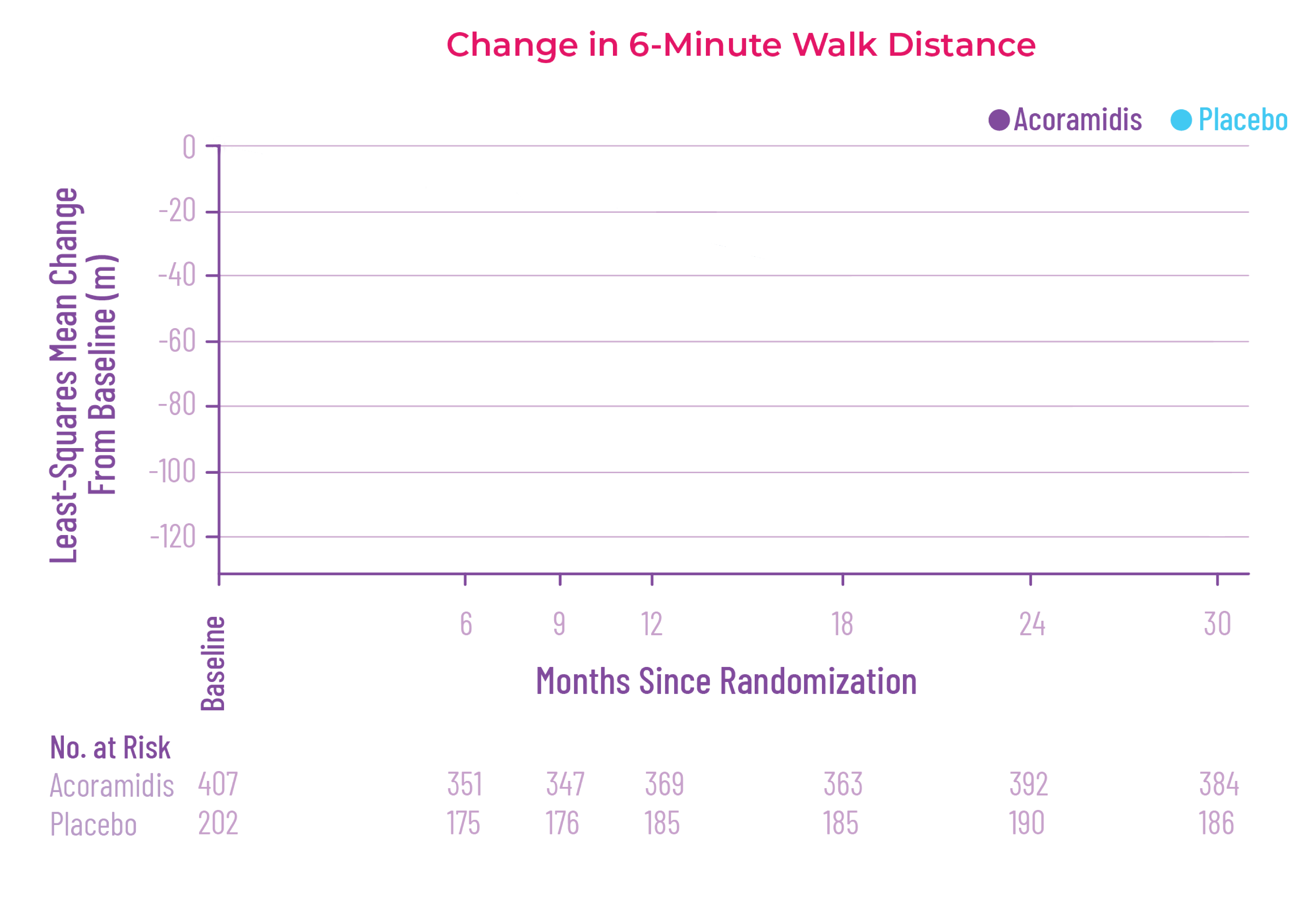
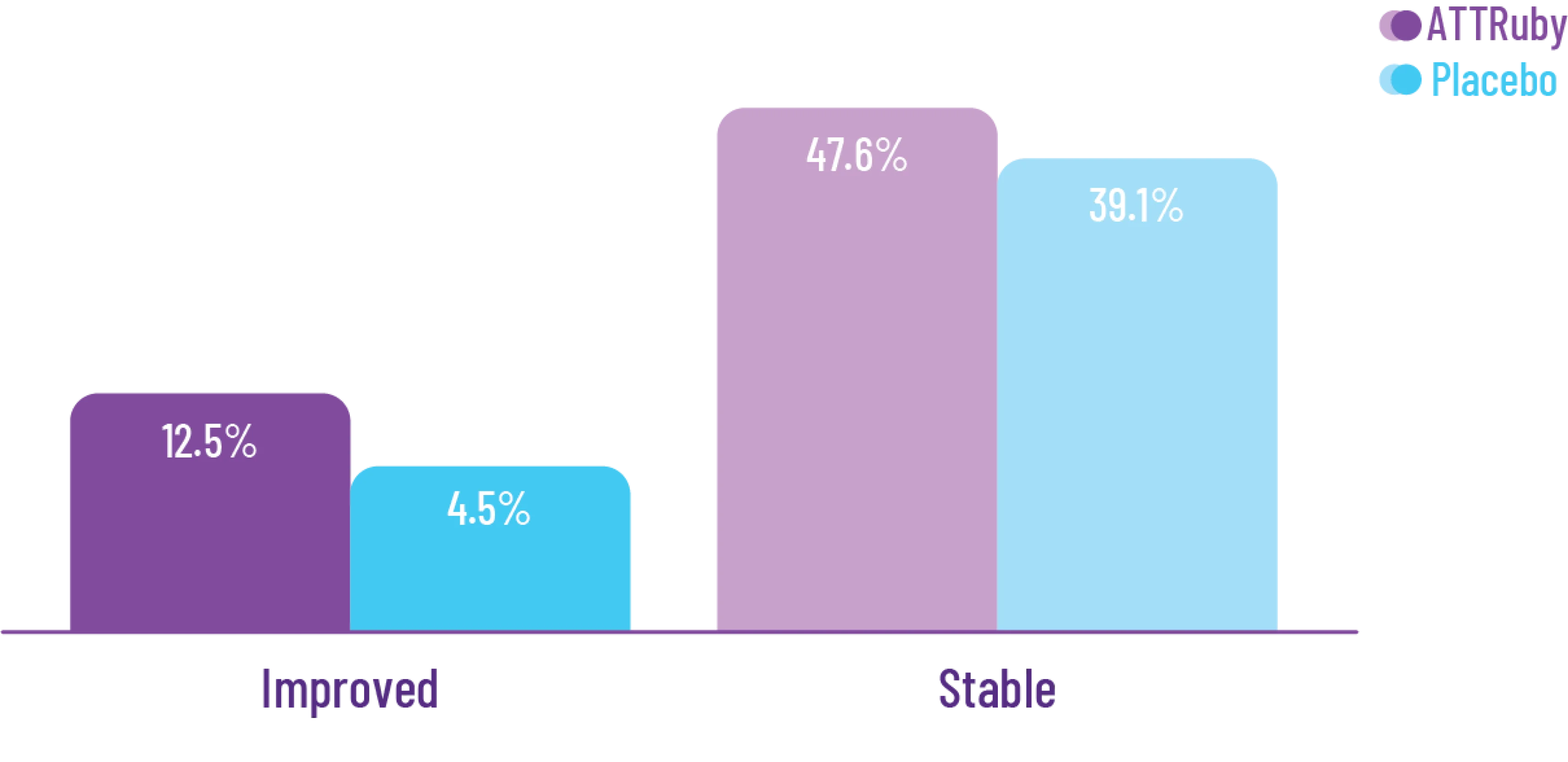
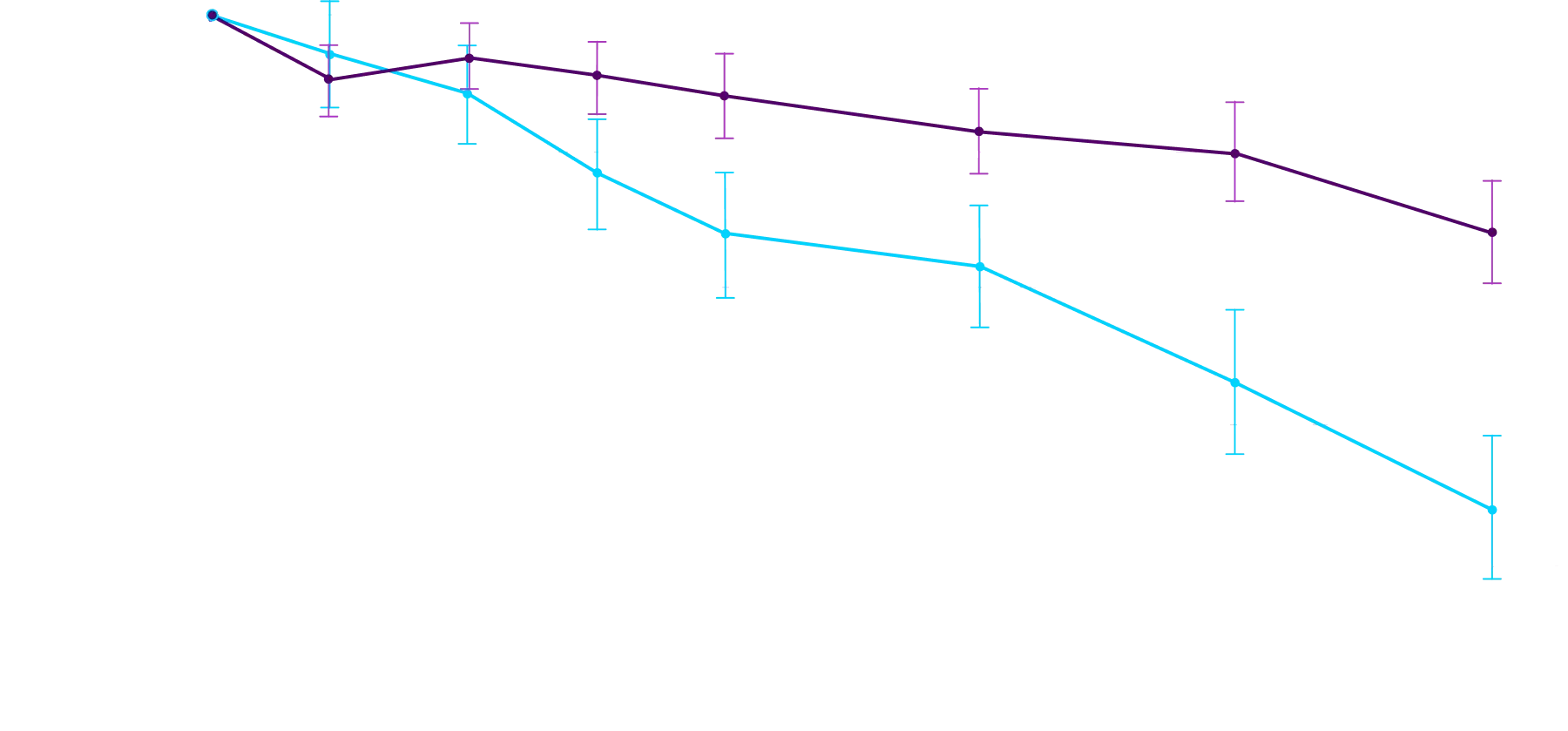
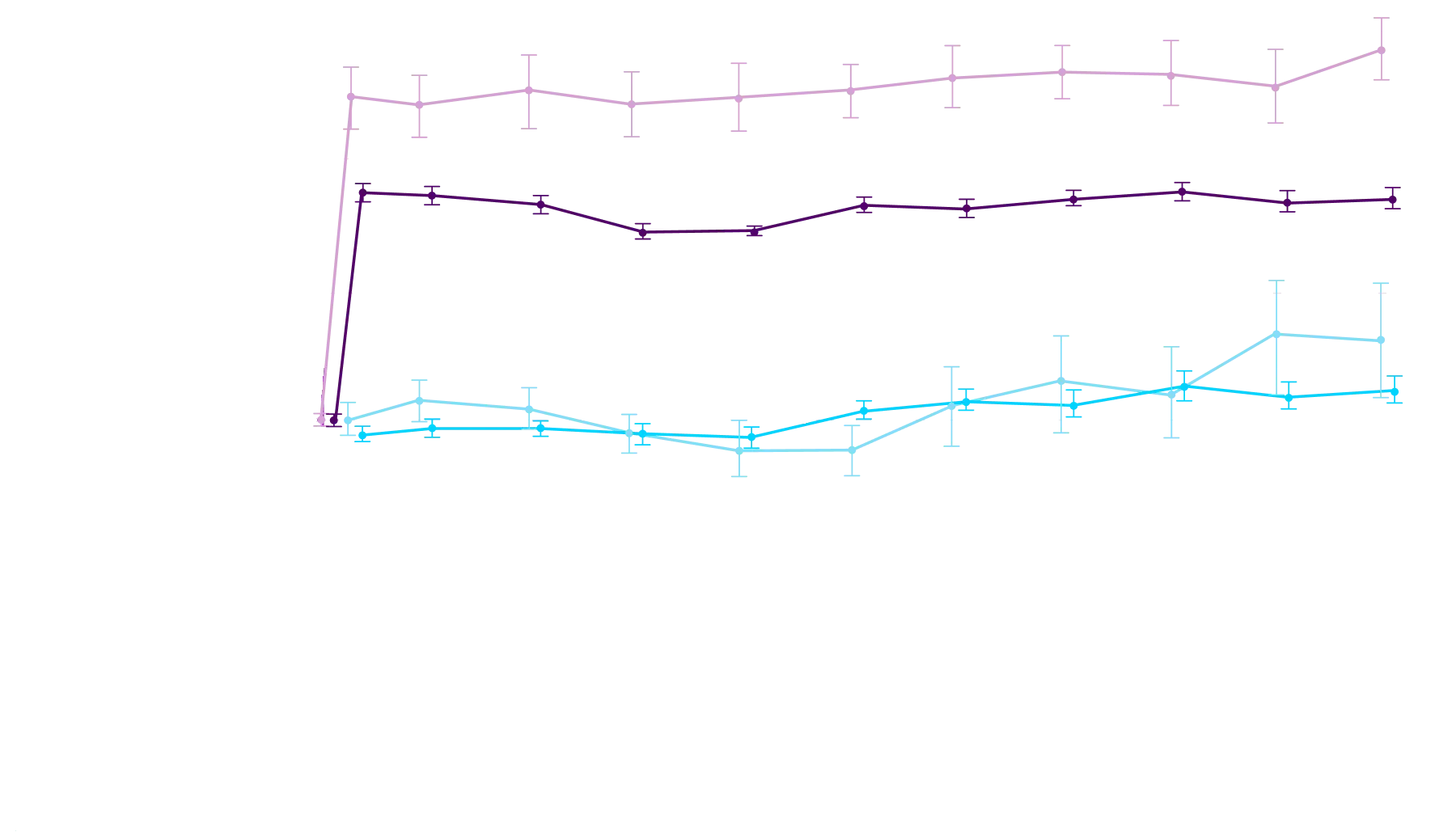
At 30 months, patients treated with
Attruby had better 6MWD results
than those who were not treated
with Attruby1
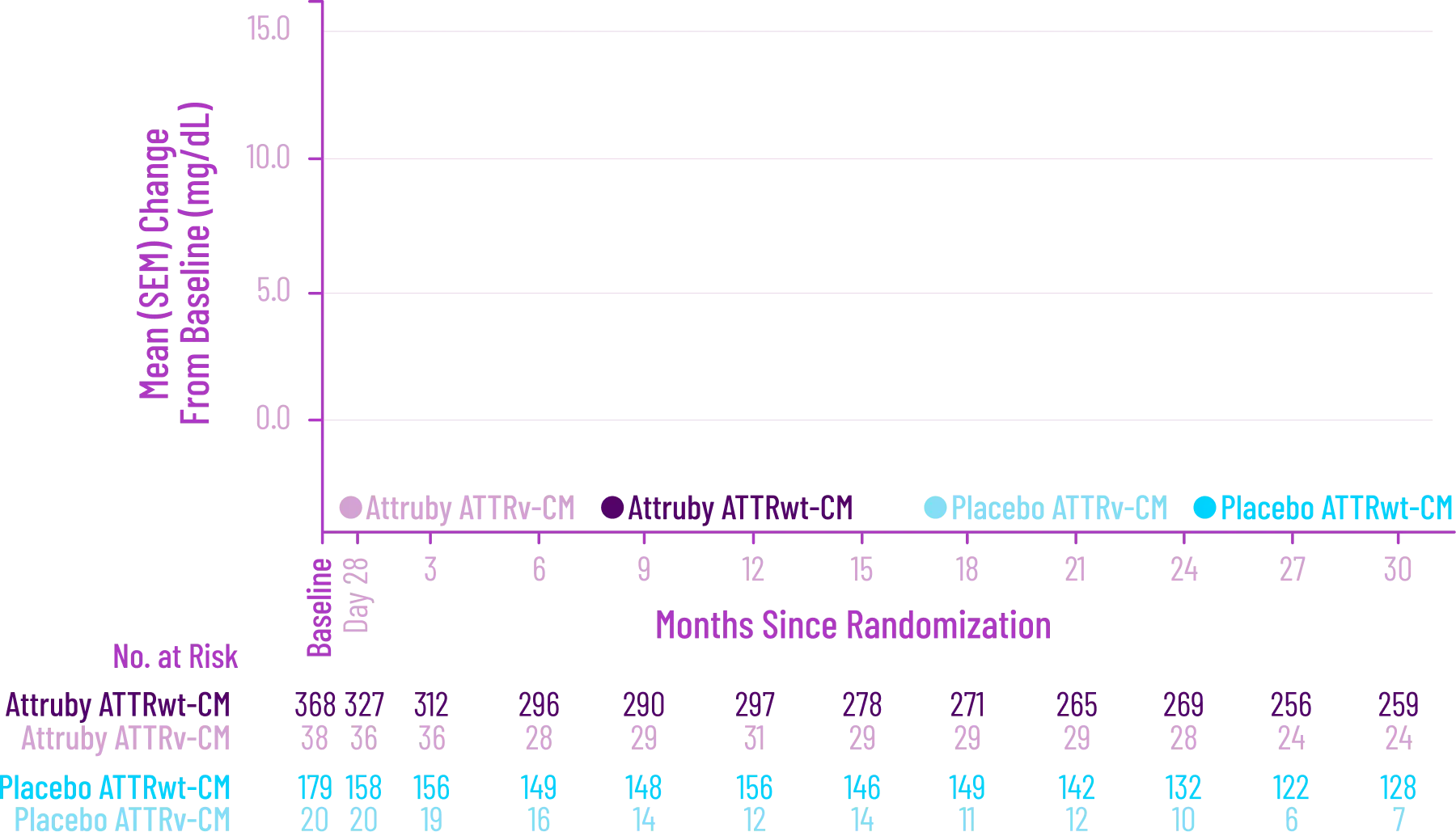
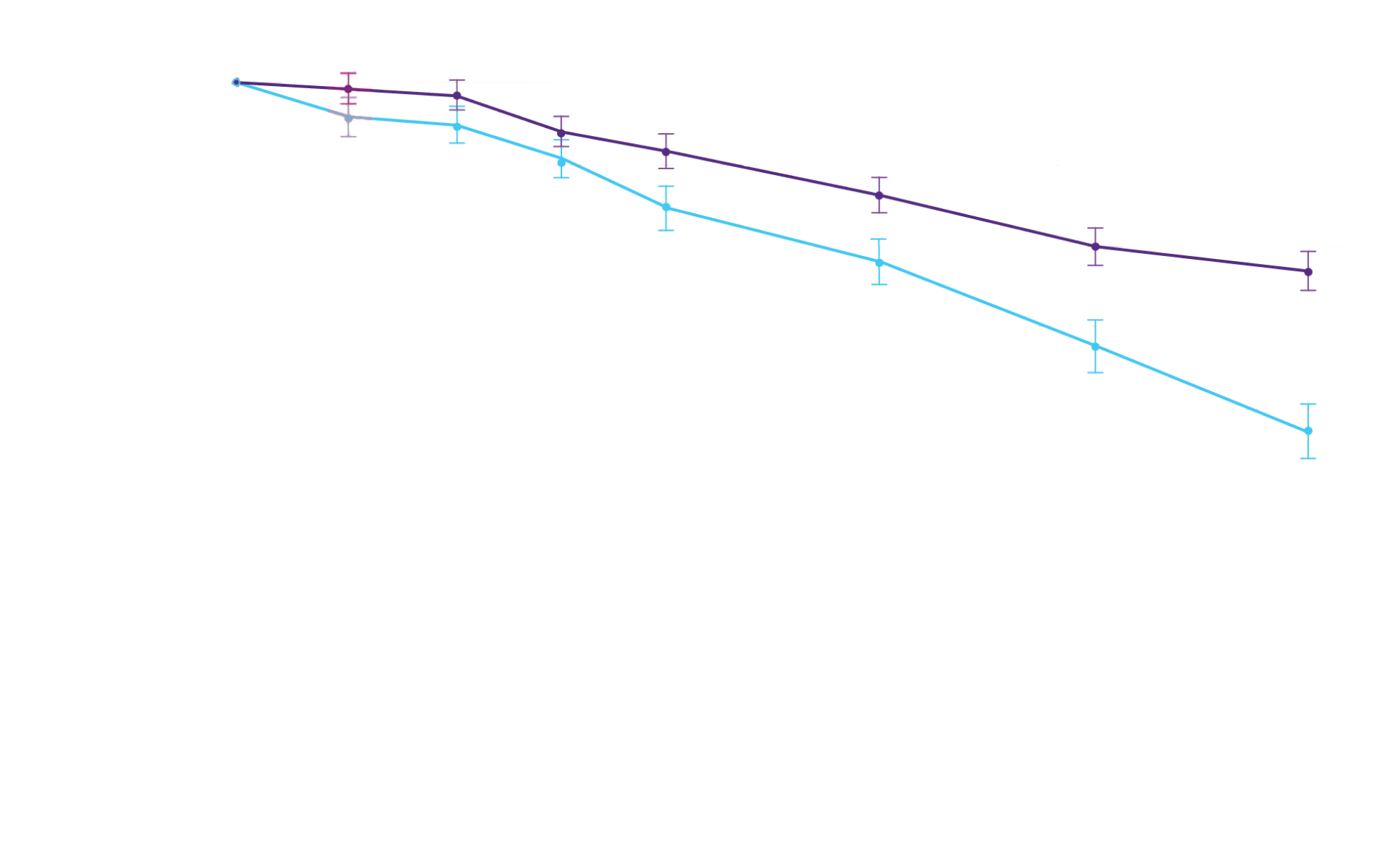
Key Secondary Endpoint:
Change From Baseline in 6MWD1
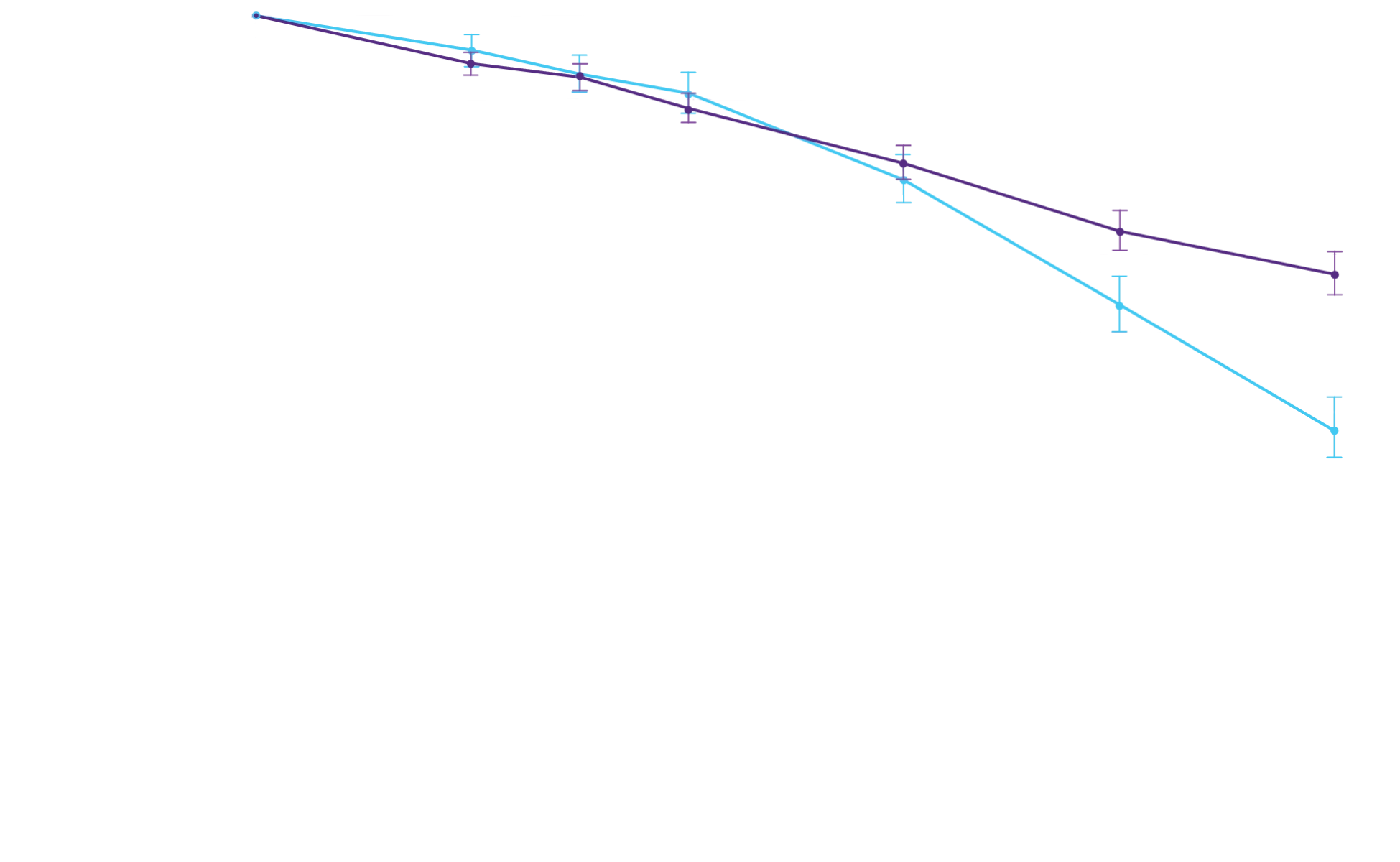
6MWD change from baseline
(95% CI: 69-190; P<0.0001)‡
†Some studies have shown that a mean 5-point change reflects a clinically meaningful change in health status.3
‡Equivalent to a difference of 40 meters. Analyzed using MMRM.4
6MWD=6-minute walk distance; KCCQ-OS=Kansas City Cardiomyopathy Questionnaire Overall Summary; LS mean=least-squares mean; MMRM=mixed model repeated measures.
References: 1. Attruby. Prescribing information. BridgeBio Pharma, Inc.; 2024.
2. Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2):132-142. doi:10.1056/NEJMoa2305434
3. Spertus JA, Jones PG, Sandhu AT, Arnold SV.
Interpreting the Kansas City Cardiomyopathy Questionnaire in clinical trials and clinical care: JACC state-of-the-art review. J Am Coll Cardiol. 2020;76(20): 2379-
2390. doi:10.1016/j.jacc.2020.09.542
4. Data on file. BridgeBio Pharma, Inc.; 2024-2025.
5. Fontana M, Kumar V, Sheridan P, et al. Descriptive analysis of unmet need
in a contemporary cohort of tafamidis-treated patients with ATTR-CM. Poster presented at: Heart Failure Society of America Annual Scientific Meeting;
September 27-30, 2024; Atlanta, GA.