- The V30M mutation can induce structural alterations that lead to amyloid deposition. Researchers found that the T119M mutation may mitigate the pathogenic effects of the V30M mutation
- Carriers of T119M had higher levels of serum TTR due to slower clearance of the protein from serum, as assessed by clearance studies
Inspired by nature. Designed with purpose.
Watch >90%
Stabilization in
Action

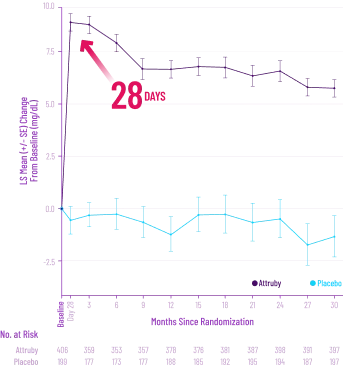
Low rates of discontinuations due to adverse drug reactions and convenient dosing with Attruby6,7
See Dosing + SafetyATTR-CM=transthyretin amyloid cardiomyopathy; TTR=transthyretin.
References: 1. Liz MA, Coelho T, Bellotti V, Fernandez-Arias MI, Mallaina P, Obici L. A narrative review of the role of transthyretin in health and disease. Neurol Ther. 2020;9(2):395-402. doi:10.1007/s40120-020-00217-0 2. Vieira M, Saraiva MJ. Transthyretin: a multifaceted protein. Biomol Concepts. 2014;5(1):45-54. doi:10.1515/bmc-2013-0038 3. Ruberg FL, Grogan M, Hanna M, Kelly JW, Maurer MS. Transthyretin amyloid cardiomyopathy: JACC state-of-the-art review. J Am Coll Cardiol. 2019;73(22):2872-2891. doi:10.1016/j.jacc.2019.04.003 4. Kittleson MM, Maurer MS, Ambardekar AV, et al. Cardiac amyloidosis: evolving diagnosis and management: a scientific statement from the American Heart Association. Circulation. 2020;142(1):e7-e22. doi:10.1161/CIR.0000000000000792 5. Witteles RM, Bokhari S, Damy T, et al. Screening for transthyretin amyloid cardiomyopathy in everyday practice. JACC Heart Fail. 2019;7(8):709-716. doi:10.1016/j.jchf.2019.04.010 6. Attruby. Prescribing information. BridgeBio Pharma, Inc.; 2024. 7. Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2)(Suppl 1):1-34. doi:10.1056/NEJMoa2305434 8. Gertz MA, Benson MD, Dyck PJ, et al. Diagnosis, prognosis, and therapy of transthyretin amyloidosis. J Am Coll Cardiol. 2015;66(21):2451-2466. doi:10.1016/j.jacc.2015.09.075 9. Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2):132-142. doi:10.1056/NEJMoa2305434 10. Hammarström P, Jiang X, Hurshman AR, Powers ET, Kelly JW. Sequence-dependent denaturation energetics: a major determinant in amyloid disease diversity. Proc Natl Acad Sci USA. 2002;99(suppl 4):16427-16432. doi:10.1073/pnas.202495199 11. Almeida ZL, Vaz DC, Brito RMM. Transthyretin mutagenesis: impact on amyloidogenesis and disease. Crit Rev Clin Lab Sci. 2024;61(7):616-640. doi:10.1080/10408363.2024.2350379 12. Hornstrup LS, Frikke-Schmidt R, Nordestgaard BG, Tybjaerg-Hansen A. Genetic stabilization of transthyretin, cerebrovascular disease, and life expectancy. Arterioscler Thromb Vasc Biol. 2013;33(6):1441-1447. doi:10.1161/ATVBAHA.113.301273 13. Judge DP, Heitner SB, Falk RH, et al. Transthyretin stabilization by AG10 in symptomatic transthyretin amyloid cardiomyopathy. J Am Coll Cardiol. 2019;74(3):285-295. doi:10.1016/j.jacc.2019.03.012 14. Maurer MS, Judge DP, Gillmore JD, et al. Early increase in serum transthyretin by acoramidis independently predicts improved survival in TTR amyloid cardiomyopathy. J Am Coll Cardiol. 2025;85(20):1911-1923. doi:10.1016/j.jacc.2025.03.542 15. Sarswat N, Ambardekar A, Taubel J, et al. Acoramidis-mediated early increase in serum transthyretin is associated with lower cardiovascular-related hospitalizations and mortality: insights from the ATTRibute-CM study. Poster presented at: American College of Cardiology Annual Scientific Meeting; March 29-31, 2025; Chicago, IL. 16. Data on file. BridgeBio, Inc.; 2024-2025.
Indication and Important safety information
INDICATION
Attruby® (acoramidis) is indicated for the treatment of the cardiomyopathy of wild-type or variant transthyretin-mediated amyloidosis (ATTR-CM) in adults to reduce cardiovascular death and cardiovascular-related hospitalization.
IMPORTANT SAFETY INFORMATION
Adverse Reactions
Diarrhea (11.6% vs 7.6%) and upper abdominal pain (5.5% vs 1.4%) were
reported in patients treated with Attruby versus placebo,
respectively. The majority of these adverse reactions were mild and
resolved without drug discontinuation.
Discontinuation rates due to adverse events were similar between patients treated with Attruby versus placebo (9.3% and 8.5%, respectively).
Laboratory Tests
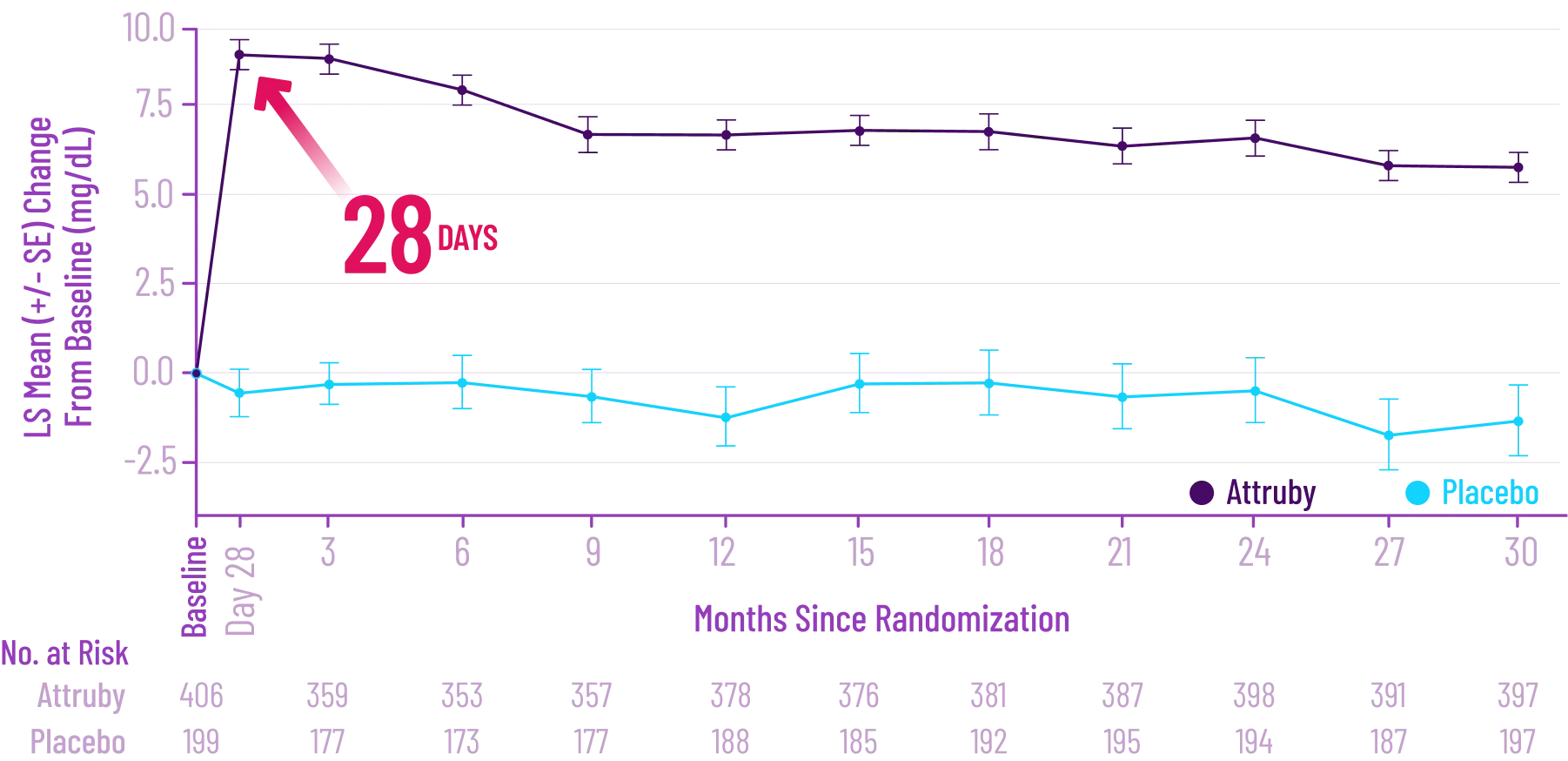
Mean increase in serum creatinine of 0.2 and 0.0 mg/dL and a mean
decrease in eGFR of 8.2 and 0.7 mL/min/1.73 m2 was
observed in the
adults with ATTR-CM treated with Attruby versus placebo,
respectively, at Day 28 and then stabilized. These changes were
reversible after treatment discontinuation.
Use in Specific Populations
Pregnancy & Lactation: There are no data on the use of Attruby in pregnant women. Animal data have not shown developmental risk associated with the use of Attruby in pregnancy. There are no available data on the presence of Attruby in either human or animal milk or the effects of the drug on the breastfed infant or maternal milk production.
Please see Full Prescribing Information including Patient Information.
INDICATION AND IMPORTANT
SAFETY INFORMATION
INDICATION
Attruby® (acoramidis) is indicated for the treatment of the cardiomyopathy of wild-type or variant transthyretin-mediated amyloidosis (ATTR-CM) in adults to reduce cardiovascular death and cardiovascular-related hospitalization.
IMPORTANT SAFETY INFORMATION
Adverse Reactions
Diarrhea (11.6% vs 7.6%) and upper abdominal pain (5.5% vs 1.4%) were
reported in patients treated with Attruby versus placebo,
respectively. The majority of these adverse reactions were mild and
resolved without drug discontinuation.
Discontinuation rates due to adverse events were similar between patients treated with Attruby versus placebo (9.3% and 8.5%, respectively).
Laboratory Tests
Mean increase in serum creatinine of 0.2 and 0.0 mg/dL and a mean
decrease in eGFR of 8.2 and 0.7 mL/min/1.73 m2 was
observed in the
adults with ATTR-CM treated with Attruby versus placebo,
respectively, at Day 28 and then stabilized. These changes were
reversible after treatment discontinuation.
Use in Specific Populations
Pregnancy & Lactation: There are no data on the use of Attruby in pregnant women. Animal data have not shown developmental risk associated with the use of Attruby in pregnancy. There are no available data on the presence of Attruby in either human or animal milk or the effects of the drug on the breastfed infant or maternal milk production.
Please see Full Prescribing Information including Patient Information.